A Levels Physics (9702)•9702/13/O/N/21
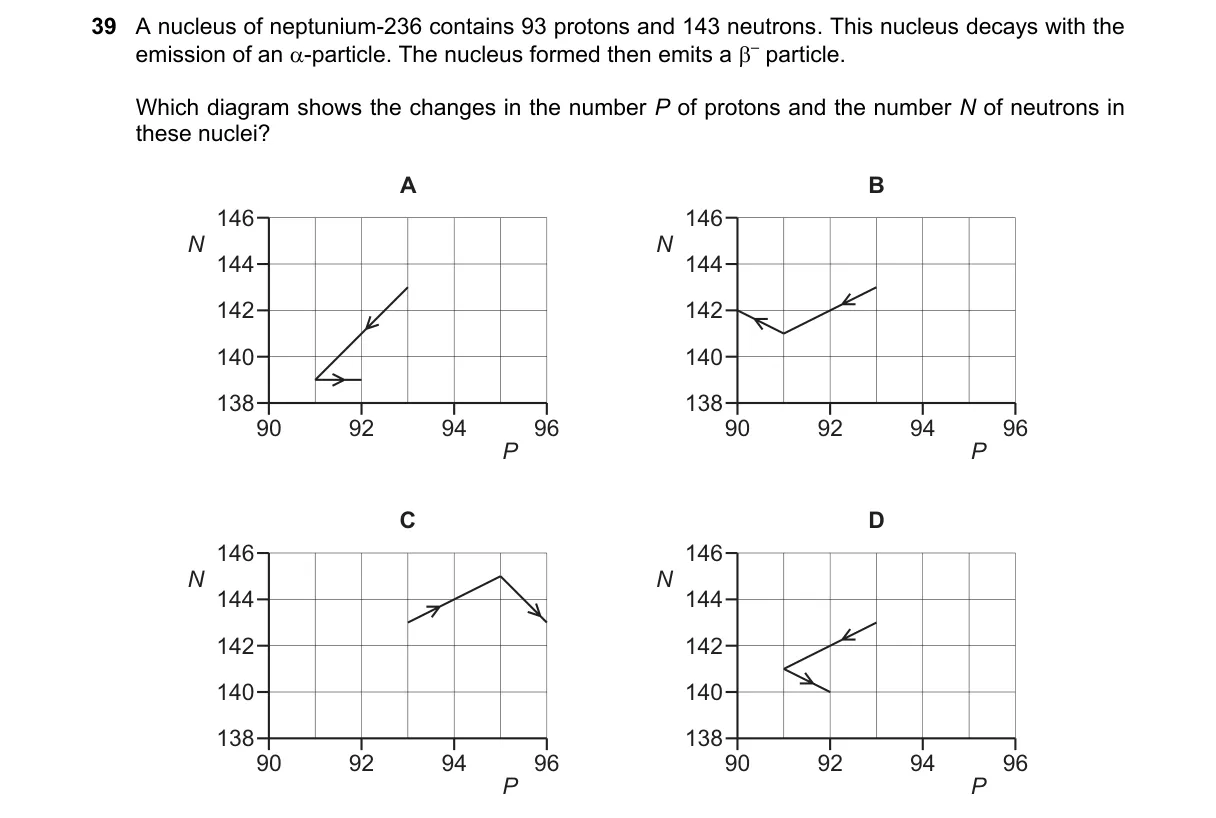
Explanation
Alpha decay emits a helium nucleus (2 protons, 2 neutrons)
Steps:
- Identify initial composition: 93 protons, 145 neutrons in neptunium-238.
- Recall alpha particle: 2 protons + 2 neutrons.
- Subtract from parent: new protons = 93 - 2 = 91; new neutrons = 145 - 2 = 143.
- Diagram must show both numbers decreasing by 2.
Why D is correct:
- D depicts protons and neutrons both reduced by 2, matching alpha emission definition.
Why the others are wrong:
- A: Shows only protons decreasing, ignoring neutron loss.
- B: Shows neutrons decreasing but protons unchanged, violating charge conservation.
- C: Shows increases in both, opposite of decay process.
Final answer: D
Topic: Radioactive decay
Practice more A Levels Physics (9702) questions on mMCQ.me