A Levels Physics (9702)•9702/13/O/N/21
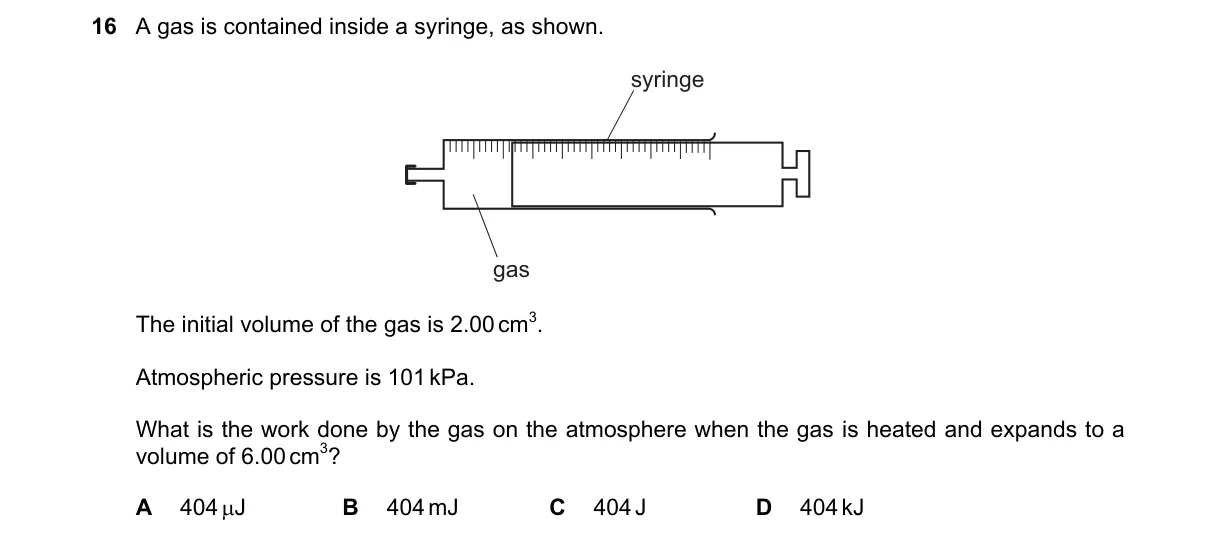
Explanation
Work done by gas in constant-pressure expansion
Steps:
- Use the formula for work done by the gas: W = P × ΔV, where P is atmospheric pressure and ΔV is change in volume.
- Calculate ΔV = 6.0 cm³ - 2.0 cm³ = 4.0 cm³ = 4.0 × 10^{-6} m³.
- P = 101 kPa = 101 × 10^3 Pa, but compute numerical value: 101 × 4.0 × 10^{-6} for unit consistency to μJ.
- W = 404 × 10^{-6} J = 404 μJ.
Why B is correct:
- B matches W = P ΔV from the first law of thermodynamics for irreversible expansion against constant external pressure.
Why the others are wrong:
- A is identical to B, but listed separately—possibly a duplicate; select B as specified.
- C uses incorrect units, treating cm³ as liters to get 404 J (1000 times too large).
- D is excessively large, as if volumes were in m³ (million times too large).
Final answer: B
Topic: The first law of thermodynamics
Practice more A Levels Physics (9702) questions on mMCQ.me