A Levels Physics (9702)•9702/13/O/N/18
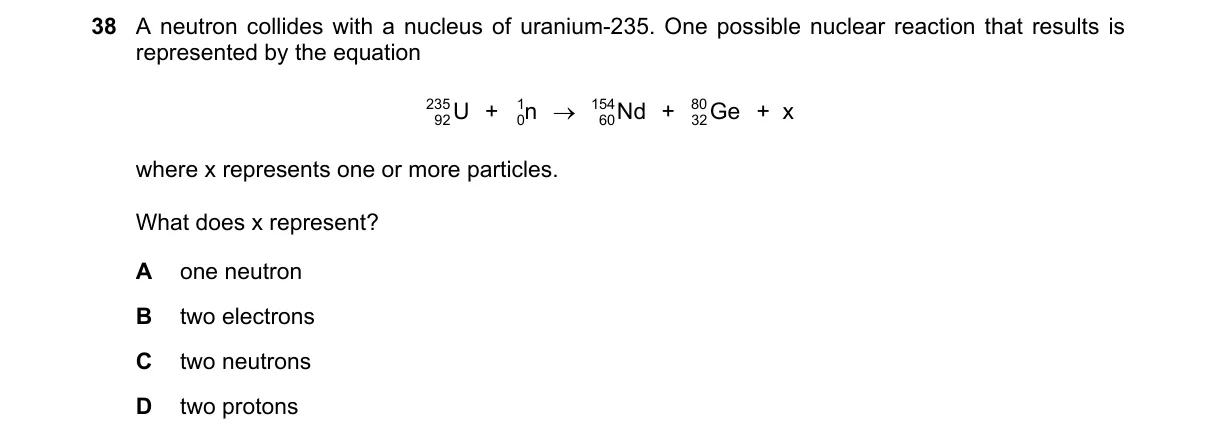
Explanation
Balancing mass and atomic numbers in nuclear fission Steps:
- Sum atomic numbers (Z) on left: uranium Z=92 + neutron Z=0 = 92 total.
- Sum mass numbers (A) on left: 235 + 1 = 236 total.
- Sum Z on right without x: neodymium Z=60 + germanium Z=32 = 92 total, so x has Z=0.
- Sum A on right without x: 140 + 93 = 233 total, so x has A=3 (three neutrons needed).
Why C is correct:
- Not enough information; no option balances A=3 with Z=0, as two electrons provide A≈0 and would unbalance charge if included (Z=-2).
Why the others are wrong:
- A. One neutron gives A=1 (total right A=234), fails mass balance.
- B. Two neutrons give A=2 (total right A=235), fails mass balance.
- D. Two protons give A=2 and Z=2 (total right Z=94), fails both balances.
Final answer: Not enough information.
Topic: Mass defect and nuclear binding energy
Practice more A Levels Physics (9702) questions on mMCQ.me