A Levels Chemistry (9701)•9701/12/O/N/24
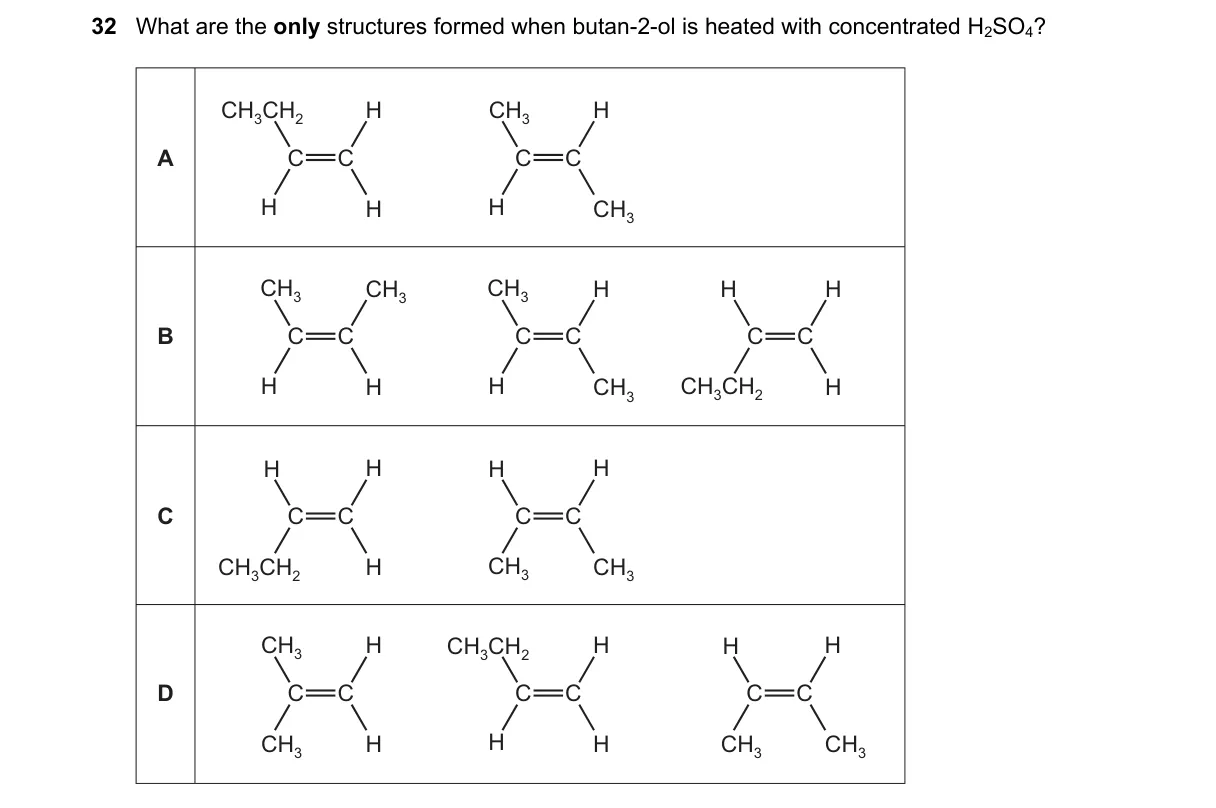
Explanation
Dehydration of but-2-ol via E1 mechanism
Steps:
- But-2-ol (CH3CH(OH)CH2CH3) protonates with H2SO4, loses water to form secondary carbocation at C2.
- Carbocation eliminates H+ from adjacent beta-carbons (C1 or C3).
- Elimination from C1 (CH3 group) yields but-1-ene: CH3CH2CH=CH2.
- Elimination from C3 (CH2 group) yields but-2-ene: CH3CH=CHCH3.
Why B is correct:
- Products match Zaitsev's rule, favoring more substituted alkene (but-2-ene) alongside minor but-1-ene from beta-elimination.
Why the others are wrong:
- A: Lists propene (CH3CH=CH2), requiring invalid C-C bond cleavage.
- C: Identical to A, includes incorrect propene.
- D: Includes butane (CH3CH2CH3), an alkane not formed in dehydration.
Final answer: B
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me