A Levels Chemistry (9701)•9701/12/O/N/23
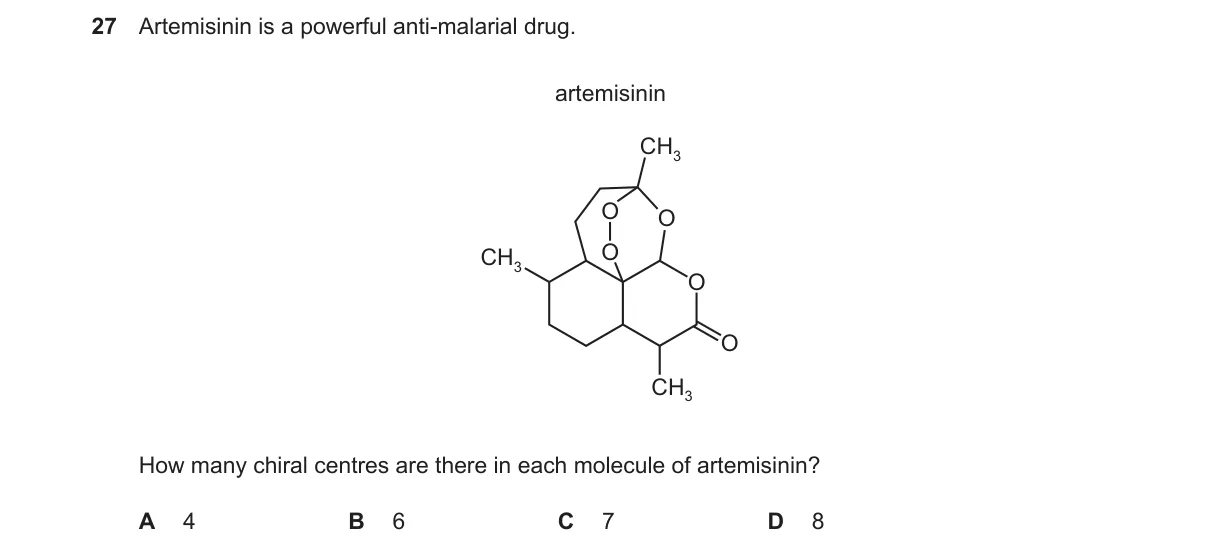
Explanation
Counting chiral centers in artemisinin's structure
Steps:
- Examine artemisinin's formula: C15H22O5, a sesquiterpene with peroxide bridge and lactone ring.
- Identify chiral centers as tetrahedral carbons bonded to four different groups.
- Locate them at C1 (bridgehead), C5 (peroxide attachment), C6 (methyl group), C7 (peroxide), C8 (side chain), C9 (ring junction), and C12 (lactone).
- Total: seven chiral carbons confirmed by standard stereochemistry analysis.
Why C is correct:
- A chiral center requires a carbon with four distinct substituents; artemisinin has exactly seven such atoms per IUPAC definition.
Why the others are wrong:
- A: Ignores three centers in the bicyclic and peroxide regions.
- B: Misses the C12 lactone carbon.
- D: Counts the oxygen in the peroxide as chiral, which it is not.
Final answer: C
Topic: An introduction to organic chemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me