A Levels Chemistry (9701)•9701/11/O/N/23
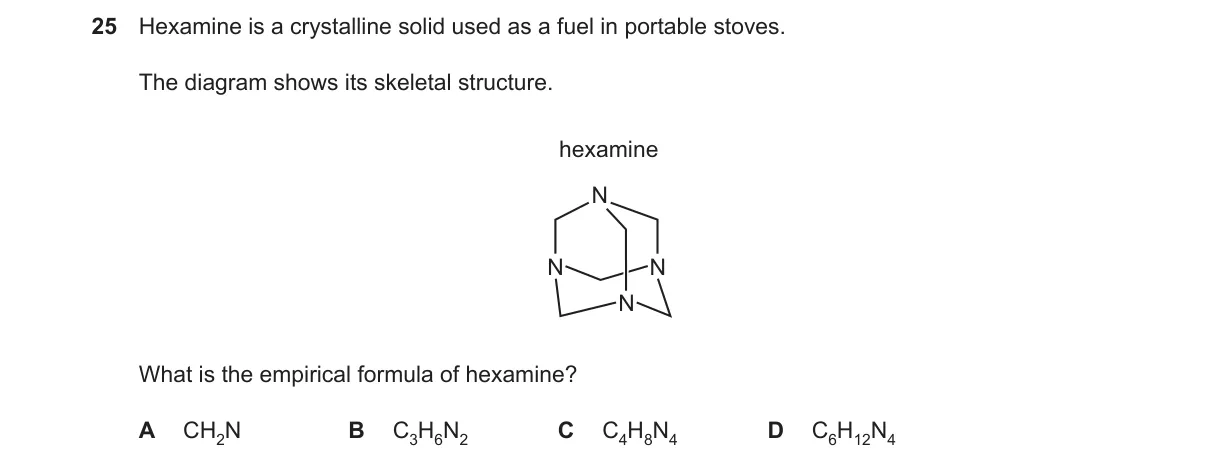
Explanation
Empirical formula from skeletal structure atom counts
Steps:
- Count carbon atoms at skeleton intersections and chain ends.
- Count explicit nitrogen atoms in the structure.
- Infer hydrogen atoms to complete valences (4 for C, 3 for N, with bonds shown or implied).
- Simplify the C:H:N ratio to lowest whole numbers.
Why B is correct:
- The structure's atom ratio simplifies to 1:4:2 per carbon, yielding CH₄N₂ as the empirical formula.
Why the others are wrong:
- A: Ratio 1:1:1 undercounts H and N relative to structure.
- C: Ratio 1:12:4 reflects molecular formula (C₆H₁₂N₄), not simplest empirical.
- D: Ratio 1:6:4 mismatches the actual simplified H and N counts.
Final answer: B
Topic: Nitrogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me