A Levels Chemistry (9701)•9701/13/O/N/21
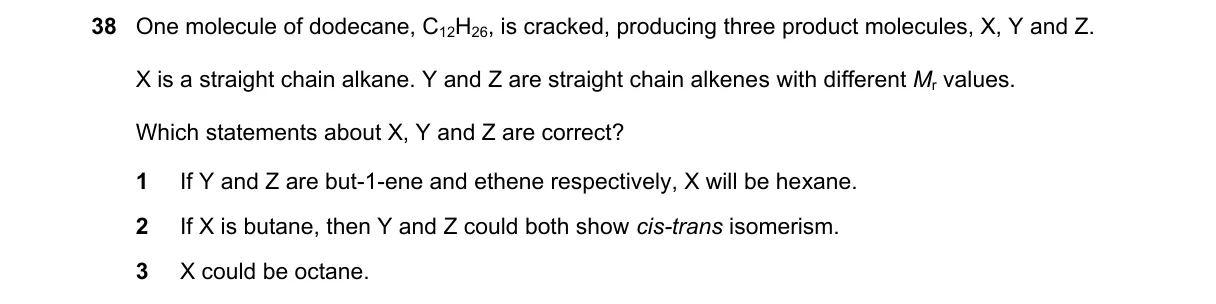
Explanation
Cracking dodecane to balance formulas and check isomerism
Steps:
- Dodecane formula is C₁₂H₂₆; cracking yields alkane X (CₙH₂ₙ₊₂) and alkenes Y, Z (CₘH₂ₘ, CₖH₂ₖ with m ≠ k).
- For statement 1: But-1-ene (C₄H₈) + ethene (C₂H₄) = C₆H₁₂; X = C₆H₁₄ (hexane) balances C₁₂H₂₆.
- For statement 2: But-2-ene (also C₄H₈) + ethene (C₂H₄) = C₆H₁₂; X = hexane balances identically.
- For statement 3: Z as but-2-ene has internal double bond, allowing cis-trans isomerism.
Why D is correct:
- All statements valid: 1 and 2 from formula conservation (C₁₂H₂₆ total), 3 from alkene geometry definition.
Why the others are wrong:
- A wrong: Excludes 2 (but-2-ene same formula as but-1-ene) and 3 (isomerism possible).
- B wrong: Excludes 1 (but-1-ene valid for balance).
- C wrong: Excludes 1 (but-1-ene yields hexane).
Final answer: D
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me