A Levels Chemistry (9701)•9701/13/O/N/21
Explanation
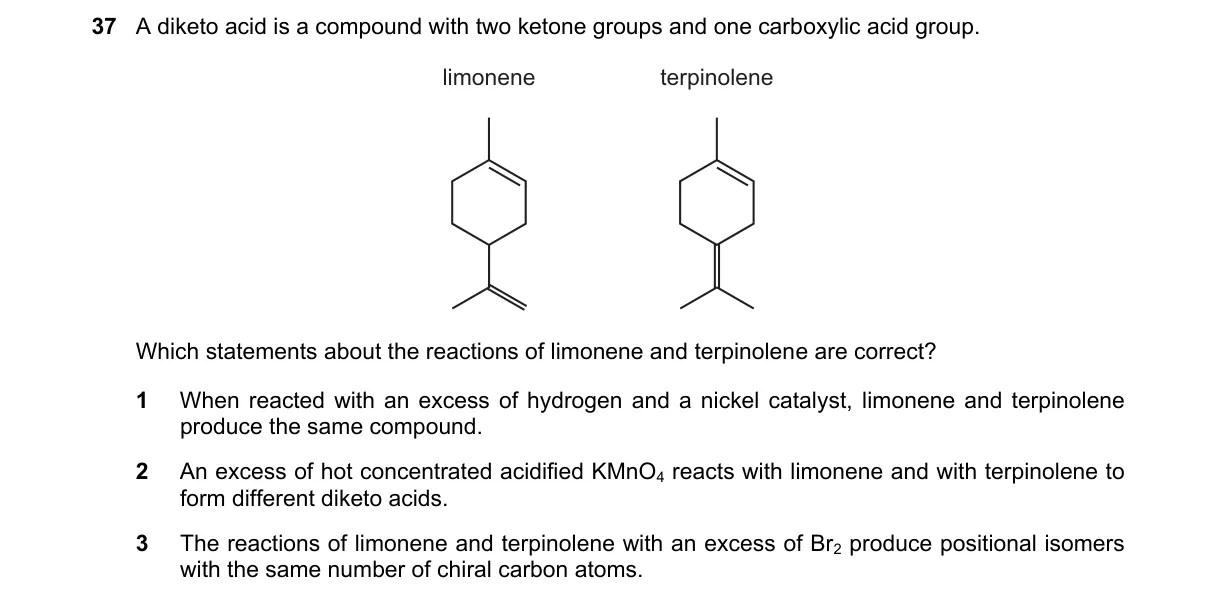
Oxidative Cleavage Differences in Terpenes
Steps:
- Limonene has an endocyclic trisubstituted alkene and an exocyclic 1-methylethenyl group; α-terpinene has conjugated dienes in the ring with methyl and isopropyl substituents.
- Hot concentrated KMnO4 cleaves C=C bonds: =CR2 → ketone, =CHR → carboxylic acid, =CH2 → CO2.
- Limonene cleavage yields a diketo acid (acetone unit becomes methyl ketone, ring opens to keto-acid chain).
- Terpinene cleavage yields a different diketo acid due to conjugated system and substituent positions, resulting in distinct carbonyl placements.
Why B is correct:
- KMnO4 oxidation cleaves double bonds based on substitution, producing unique diketo acids from limonene's isolated alkenes vs. terpinene's conjugated dienes (structural isomerism dictates product formula).
Why the others are wrong:
- A: Both hydrogenate to identical p-menthane (1-isopropyl-4-methylcyclohexane), so same compound.
- C: Br2 addition yields stereoisomers, not positional; chiral carbons vary (limonene product has 2, terpinene may differ).
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me