A Levels Chemistry (9701)•9701/13/O/N/21
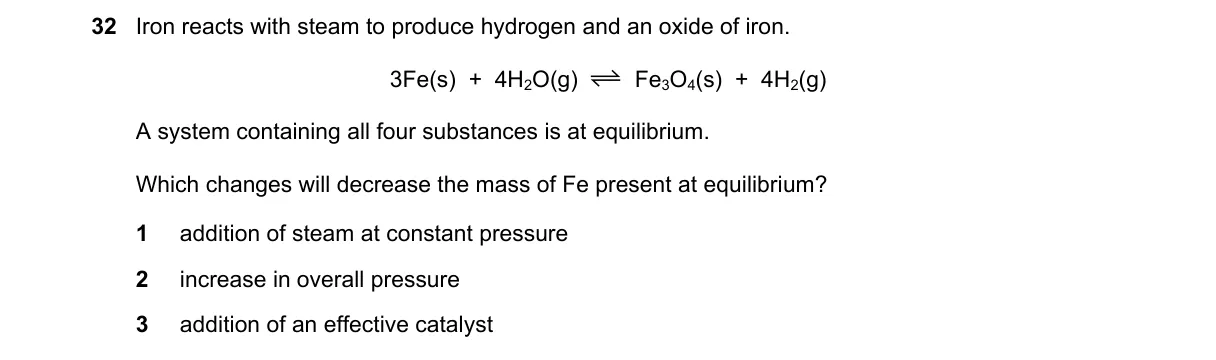
Explanation
Le Chatelier's principle on heterogeneous equilibrium
Steps:
- Identify the equilibrium: 3Fe(s) + 4H₂O(g) ⇌ Fe₃O₄(s) + 4H₂(g); Kp = (P_{H₂})⁴ / (P_{H₂O})⁴.
- Mass of Fe changes if equilibrium shifts, consuming or producing solid Fe.
- Evaluate A: Adding H₂O at constant P increases P_{H₂O}, shifting right to consume Fe.
- Note D is unspecified, making full analysis impossible.
Why D is correct:
- Not enough information (choice D blank, cannot verify effect).
Why the others are wrong:
- A: Shifts equilibrium right, decreasing Fe mass.
- B: Δn_g = 0, so no shift; Fe mass unchanged.
- C: Catalyst equalizes rates without shifting position; Fe mass unchanged.
Final answer: Not enough information.
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me