A Levels Chemistry (9701)•9701/13/O/N/21
Explanation
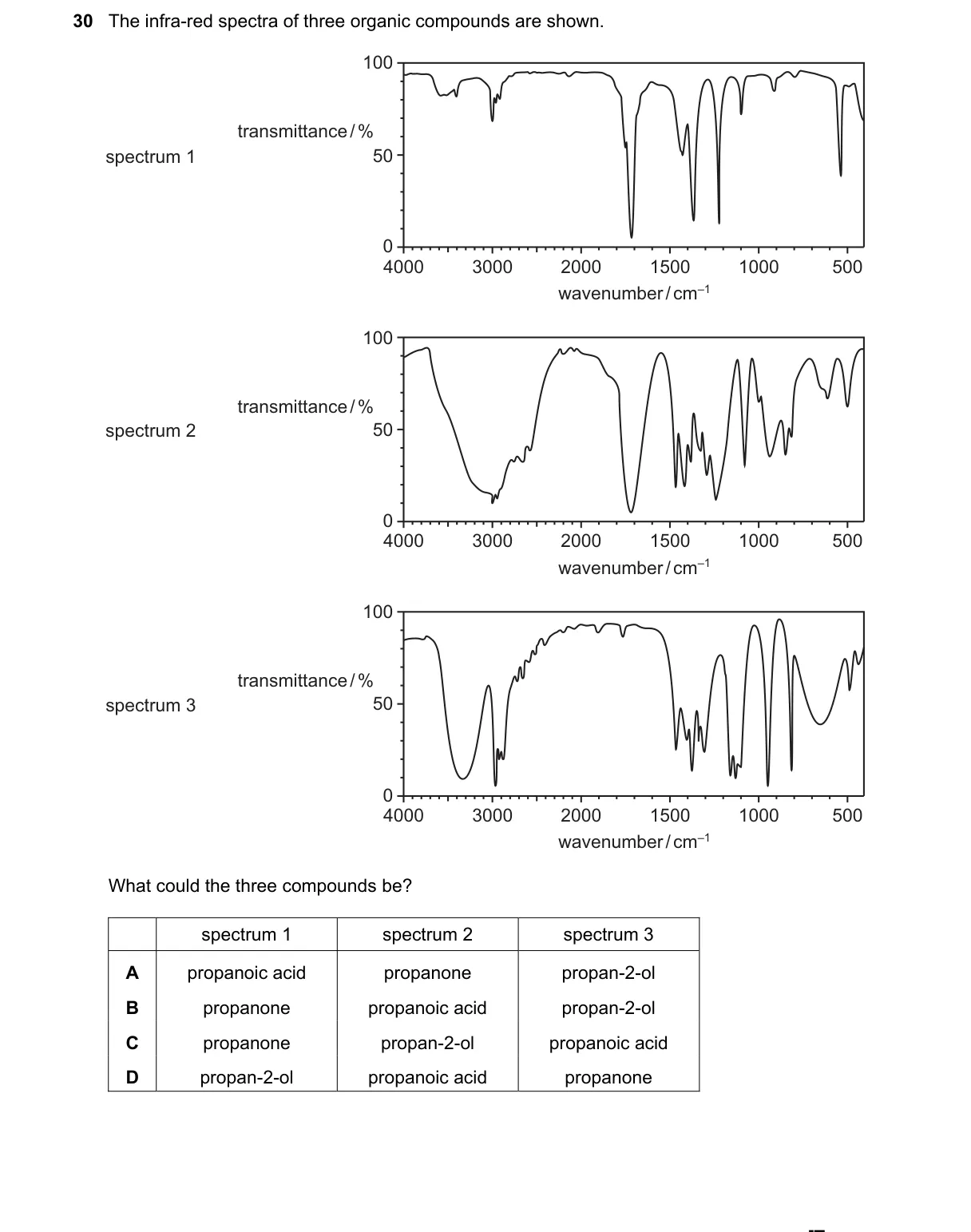
Distinguishing functional groups by IR absorption bands
Steps:
- Identify key IR features: alcohols show broad O-H at 3200-3600 cm⁻¹; carbonyls at 1700-1750 cm⁻¹; aldehydes have unique C-H at 2700-2850 cm⁻¹.
- Match spectra: one with O-H only (alcohol), one with C=O and no O-H (ketone), one with C=O, no O-H, plus aldehyde C-H (aldehyde).
- Eliminate options lacking aldehyde (unique C-H band) or mismatching alcohol type.
- Confirm B fits: propanone (ketone C=O), propanal (aldehyde C=O + C-H), propan-2-ol (secondary alcohol O-H).
Why B is correct:
- Aldehyde's characteristic C-H stretch at 2700-2850 cm⁻¹ (from sp² C-H in CHO group) uniquely identifies propanal, distinguishing it from ketone and alcohol.
Why the others are wrong:
- A lacks aldehyde; includes primary alcohol and acid, both with O-H but no aldehyde C-H.
- C includes acid (broad O-H + C=O) and primary alcohol, missing secondary alcohol distinction.
- D lacks aldehyde; has secondary alcohol and acid, but no propanal's C-H band.
Final answer: B
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me