A Levels Chemistry (9701)•9701/13/O/N/21
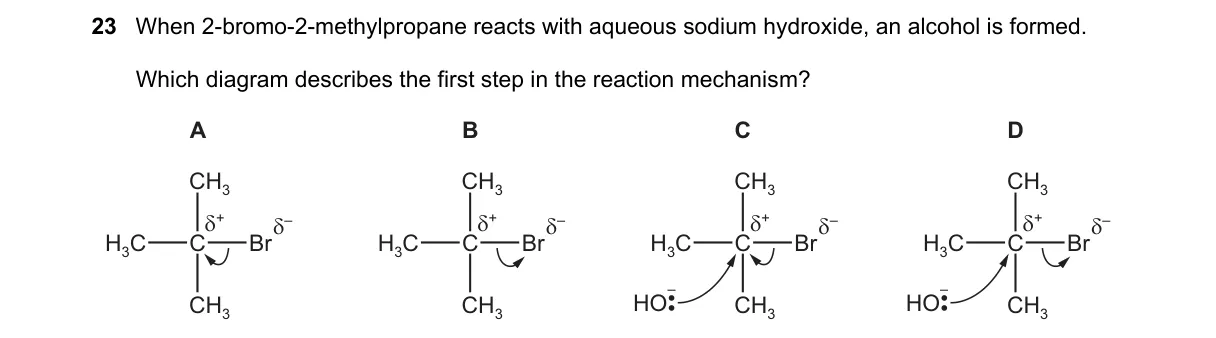
Explanation
SN1 Mechanism Initiation for Tertiary Alkyl Halide
Steps:
- Recognize 2-bromo-2-methylpropane as (CH₃)₃CBr, a tertiary alkyl halide.
- Note aqueous NaOH promotes nucleophilic substitution to form (CH₃)₃COH.
- Determine mechanism: tertiary halide in polar protic solvent favors SN1 over SN2.
- Identify first step: rate-determining heterolytic cleavage of C-Br bond, forming carbocation intermediate.
Why B is correct:
- B depicts carbocation formation via Br⁻ departure, matching SN1's unimolecular rate law (rate = k[RX]).
Why the others are wrong:
- A shows backside nucleophilic attack, characteristic of SN2 bimolecular mechanism unsuitable for tertiary halides.
- C illustrates proton abstraction, typical of E2 elimination, not substitution.
- D represents concerted OH⁻ attack with Br⁻ departure, resembling SN2, which is sterically hindered here.
Final answer: B
Topic: Halogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me