A Levels Chemistry (9701)•9701/13/O/N/21
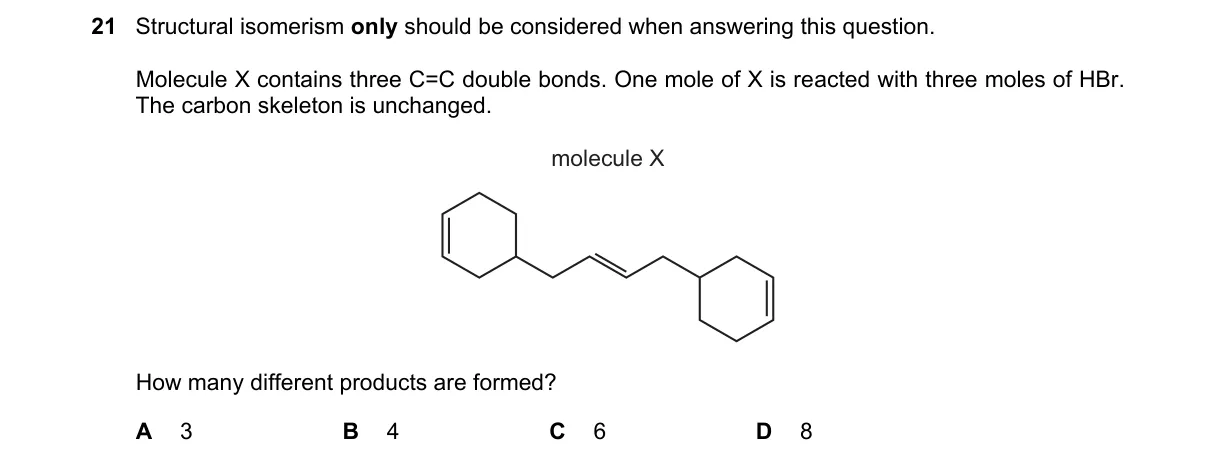
Explanation
Regioselective HBr addition to multiple unsymmetric double bonds
Steps:
- Examine the structure of X to identify the three C=C bonds and their substitution patterns.
- For each double bond, determine Markovnikov addition orientations, yielding 1 product for symmetric bonds and 2 for unsymmetric ones.
- Since the bonds are isolated (no conjugation effects mentioned), independent additions occur without skeleton change.
- Multiply regioisomers: two bonds with 2 options each, one with 1, totals 4 structural products.
Why B is correct:
- Four products arise from combinatorial regiochemistry across the three double bonds, per the definition of structural isomers as distinct connectivities.
Why the others are wrong:
- A: Underestimates by ignoring multiple unsymmetric bonds.
- C: Overcounts by including stereoisomers, excluded here.
- D: Overcounts by assuming conjugation or rearrangements, not indicated.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me