A Levels Chemistry (9701)•9701/13/O/N/21
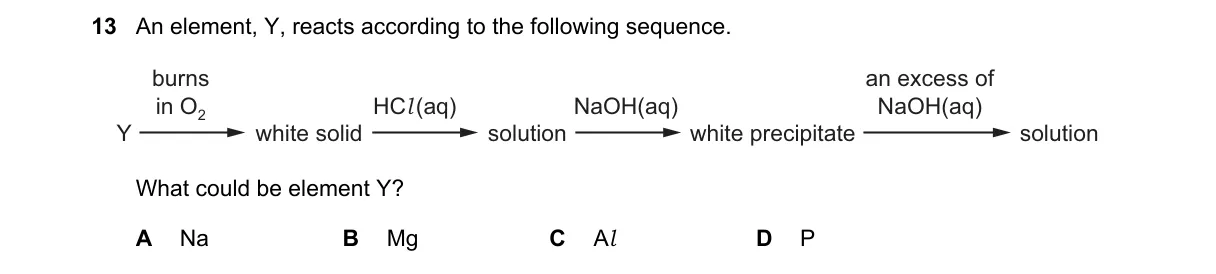
Explanation
Amphoteric oxide behavior identifies aluminum
Steps:
- Y burns in O₂ to form a white solid oxide.
- The oxide dissolves in excess HCl(aq), forming a chloride solution.
- Adding NaOH(aq) to this solution produces a white precipitate of hydroxide.
- Excess NaOH(aq) dissolves the precipitate, forming a soluble complex.
Why C is correct:
- Aluminum forms Al₂O₃ (white solid), which reacts with HCl to AlCl₃ (soluble); AlCl₃ + NaOH → Al(OH)₃ (white ppt), and excess NaOH → [Al(OH)₄]⁻ (soluble aluminate), per amphoteric hydroxide definition.
Why the others are wrong:
- A. Na: Na₂O dissolves in HCl to NaCl, but adding NaOH forms no precipitate.
- B. Mg: MgO dissolves in HCl to MgCl₂; Mg(OH)₂ precipitates with NaOH but remains insoluble in excess.
- D. P: P₄O₁₀ forms phosphoric acid with water/HCl, no hydroxide precipitate with NaOH.
Final answer: C
Topic: The Periodic Table: chemical periodicity
Practice more A Levels Chemistry (9701) questions on mMCQ.me