A Levels Chemistry (9701)•9701/13/O/N/21
Explanation
Le Chatelier's principle for exothermic equilibrium
Steps:
- The forward reaction H₂(g) + I₂(g) ⇌ 2HI(g) is exothermic (ΔH < 0).
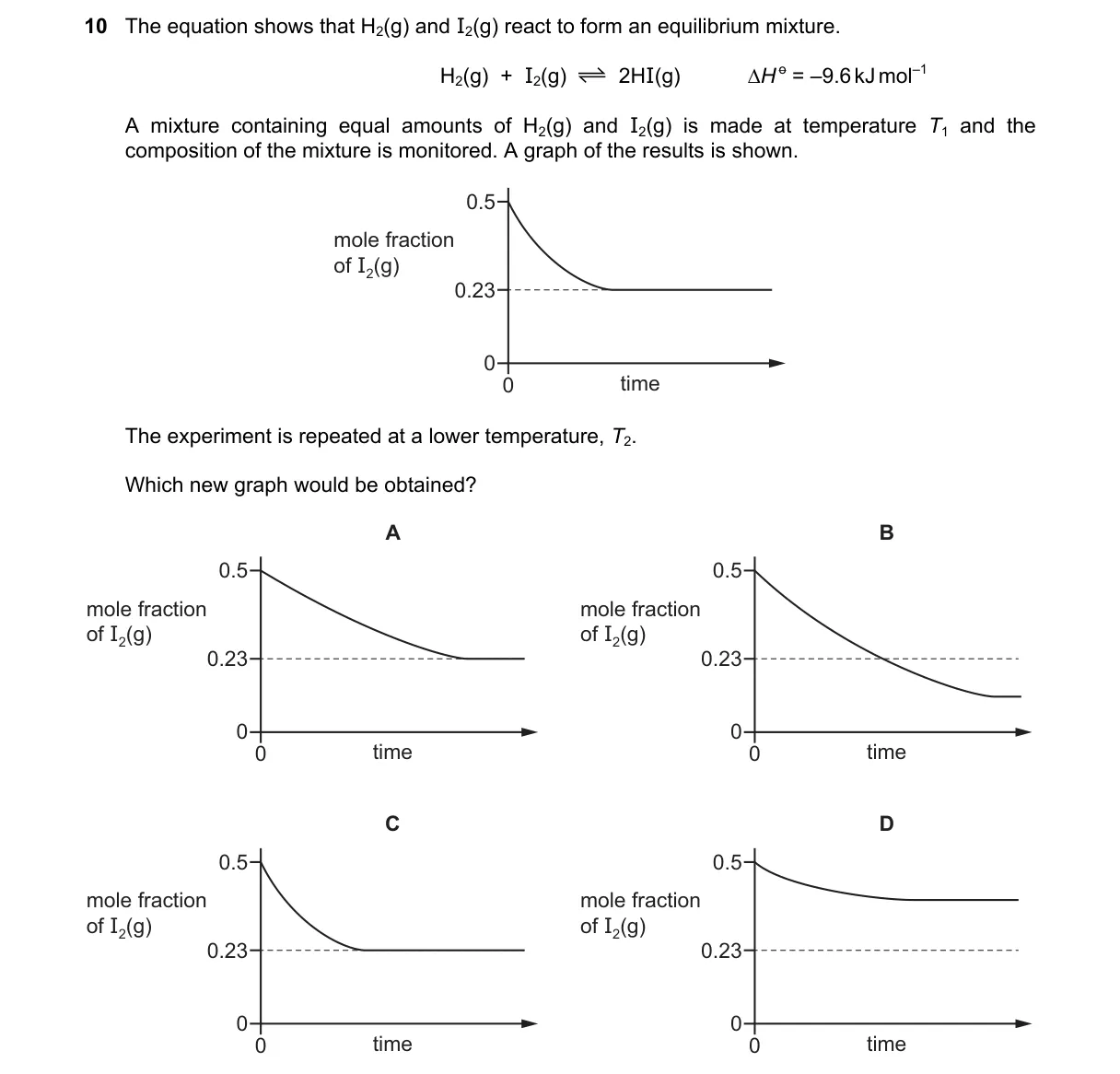
- At T₁ (500 K), the graph shows mole fraction of HI decreasing from 0.5 to 0.23 and plateauing, indicating equilibrium favors reactants due to smaller K.
- Lowering temperature to T₂ shifts equilibrium right (Le Chatelier's principle), increasing K and thus the equilibrium mole fraction of HI.
- The new graph approaches a higher HI mole fraction than 0.23 before plateauing.
Why B is correct:
- B shows the mole fraction of HI decreasing to a value above 0.23 and flattening, matching the increased K for exothermic reaction at lower T.
Why the others are wrong:
- A decreases to below 0.23 (ignores temperature effect on K).
- C shows increasing HI (inconsistent with initial conditions favoring decomposition).
- D remains flat at 0.5 (no shift toward higher product).
Final answer: B
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me