A Levels Chemistry (9701)•9701/12/O/N/21
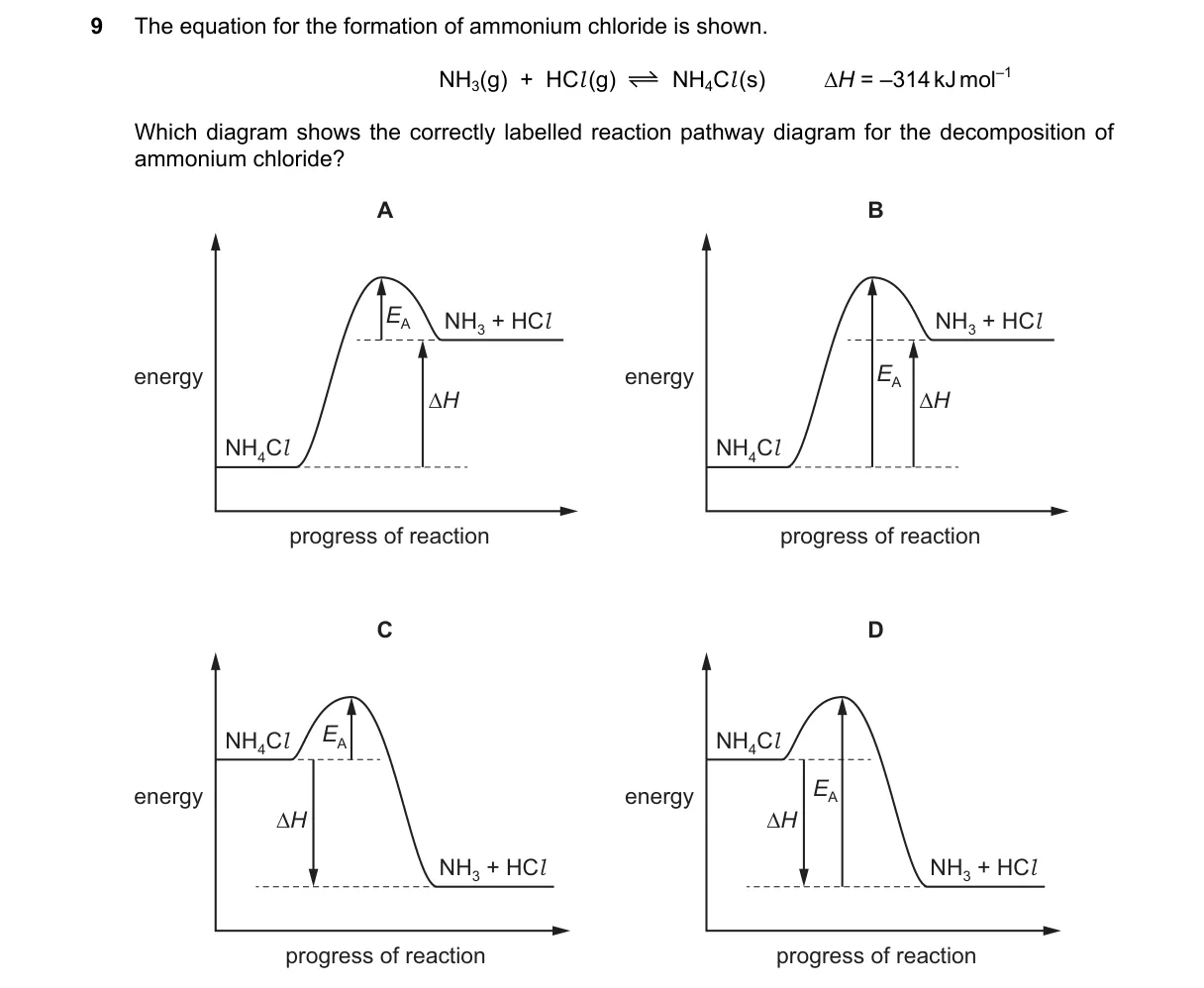
Explanation
Endothermic Decomposition Energy Diagram
Steps:
- Reverse the given exothermic formation reaction to get endothermic decomposition: NH₄Cl(s) → NH₃(g) + HCl(g), ΔH = +174 kJ mol⁻¹.
- In an energy diagram, reactants (NH₄Cl) start at lower energy level than products (gases).
- The pathway includes an activation energy barrier leading to higher product energy.
- Select diagram showing upward energy trend from reactant to products with a peak.
Why B is correct:
- Depicts reactants below products with activation energy hump, matching endothermic ΔH > 0 definition.
Why the others are wrong:
- A: Shows exothermic profile (products lower than reactants), reverse of decomposition.
- C: Lacks activation energy barrier, violating transition state theory.
- D: Inverts energy levels (products lower), incorrect for endothermic process.
Final answer: B
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me