A Levels Chemistry (9701)•9701/12/O/N/21
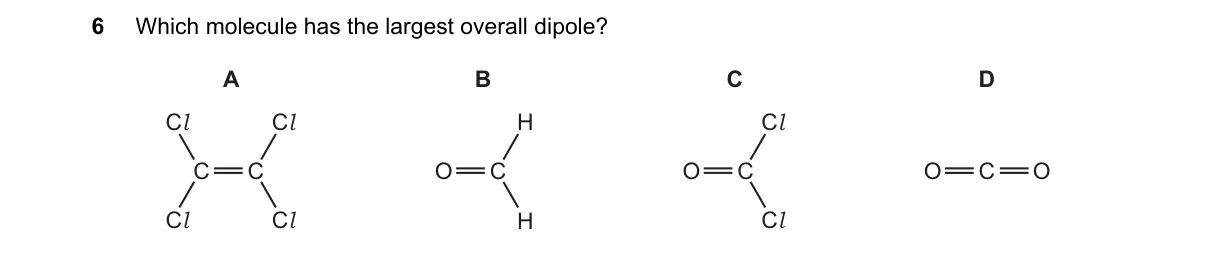
Explanation
Overall dipole moment arises from vector sum of polar bonds in asymmetric molecules
Steps:
- Identify polar bonds: C=O (electronegative O pulls electrons) in B, C, D; C-Cl in A, C.
- Assess geometry: Planar for A, B, C; linear for D.
- Check symmetry: Symmetric structures cancel dipoles; asymmetric ones retain net dipole.
- Compare net dipoles: H2C=O shows largest imbalance due to small, less electronegative H atoms.
Why B is correct:
- H2C=O features a highly polar C=O bond (electronegativity difference ΔEN=1.0) with V-shaped geometry where H atoms fail to cancel the dipole, per vector addition principle.
Why the others are wrong:
- A: Cl2C=CCl2 has symmetric trans-planar geometry, canceling C-Cl dipoles for zero net dipole.
- C: Cl2C=O has C-Cl bonds opposing the C=O dipole, reducing net moment via partial cancellation.
- D: O=C=O is linear and symmetric, with identical C=O dipoles canceling exactly.
Final answer: B
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me