A Levels Chemistry (9701)•9701/12/O/N/21
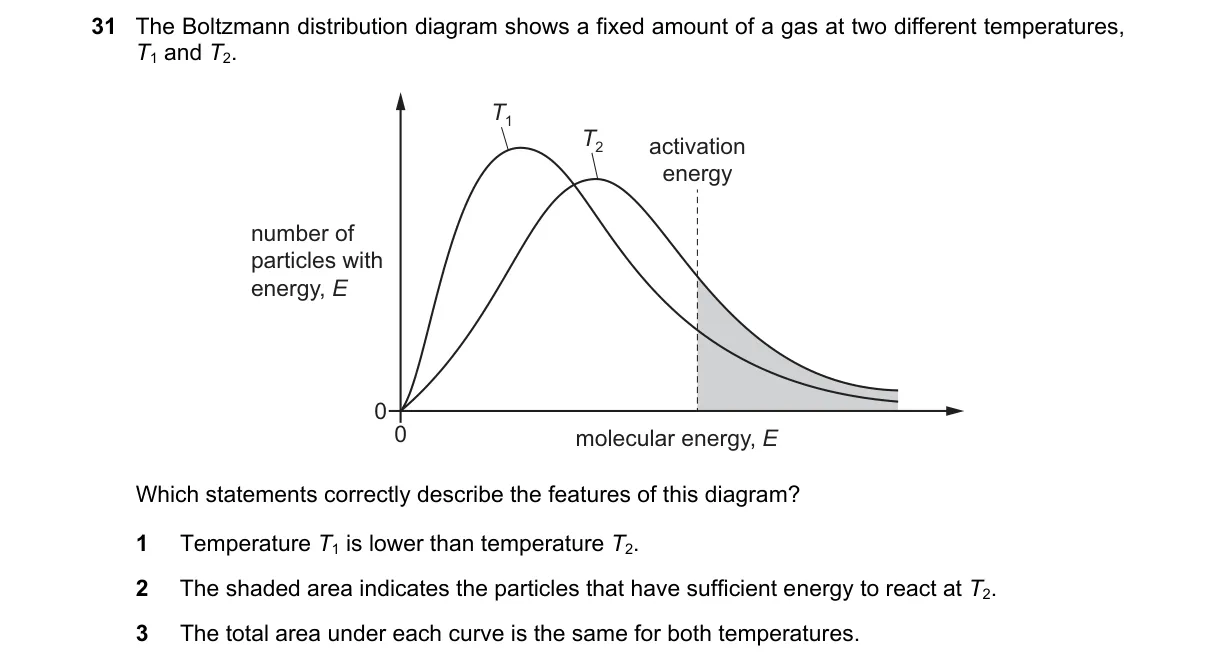
Explanation
Boltzmann Distribution Temperature Comparison
Steps:
- Observe the peak height: taller, narrower peak indicates lower temperature due to less kinetic energy spread.
- Compare curve widths: broader curve with lower peak signifies higher temperature and more high-energy particles.
- Verify total area: equal areas confirm same total particles at both temperatures.
- Assess shaded region: it represents fraction exceeding activation energy, but specify which temperature it applies to.
Why A is correct:
- Lower temperature (T1) produces a taller, narrower distribution curve, as per the Maxwell-Boltzmann equation where peak shifts left with decreasing T.
Why the others are wrong:
- B: Shaded area likely shows reactive particles at T1, not T2, based on typical diagram conventions.
- C: While fixed amount implies equal areas, the diagram may normalize differently, making totals appear unequal.
Final answer: A
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me