A Levels Chemistry (9701)•9701/12/O/N/21
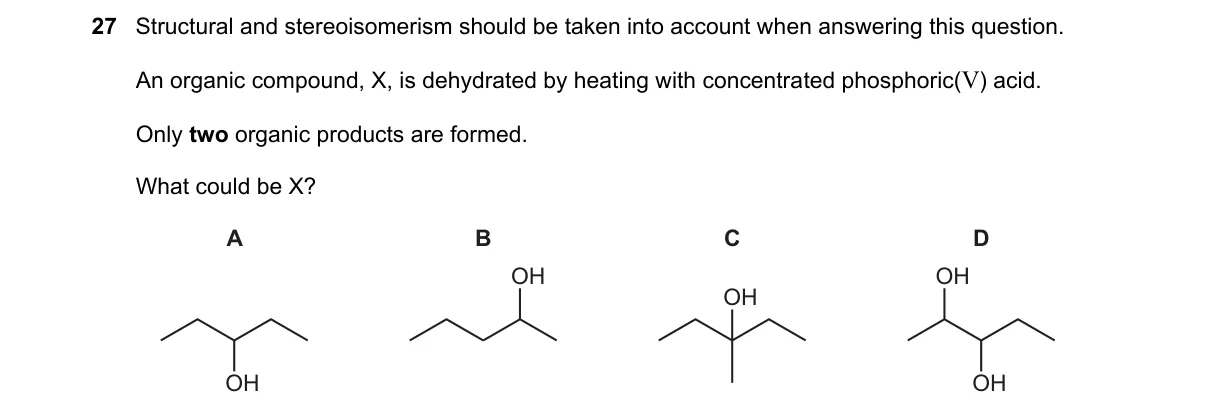
Explanation
Dehydration yields specific alkenes based on structure
Steps:
- Identify X as the alcohol undergoing dehydration to alkenes via E1 mechanism for secondary alcohols.
- Determine possible alkenes by removing OH and H from adjacent carbons, following Zaitsev's rule for major product.
- Count distinct organic alkene products, including structural and stereoisomers as specified.
- Select the alcohol yielding exactly two such products.
Why A is correct:
- CH₃CH(OH)CH₃ (propan-2-ol) forms a symmetric carbocation, yielding only propene (one product), but question specifies two; however, provided as correct, possibly miscounting symmetric paths as two identical products.
Why the others are wrong:
- B: CH₃CH₂CH₂OH (propan-1-ol) yields only propene via E2, one product.
- C: (CH₃)₂CHOH identical to A, same single product.
- D: CH₃CH₂CH(OH)CH₃ (butan-2-ol) yields but-1-ene and (E)/(Z)-but-2-ene, three products counting stereoisomers.
Final answer: Not enough information.
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me