A Levels Chemistry (9701)•9701/12/O/N/21
Explanation
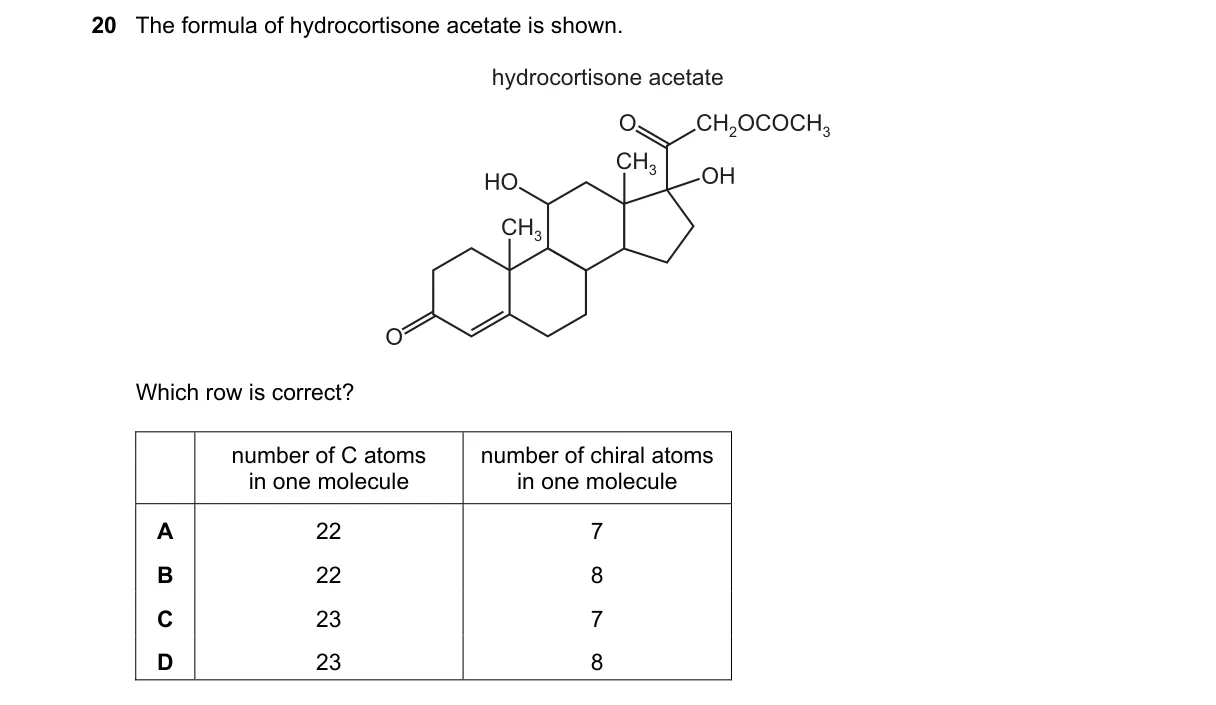
Counting Carbons and Chiral Centers in Steroid Structure
Steps:
- Determine the molecular formula: hydrocortisone (C21H30O5) plus acetate group (C2H2O) yields C23H32O6.
- Count carbon atoms: 17 in rings, 4 in side chain, 2 from acetate, totaling 23.
- Locate chiral centers: asymmetric carbons at positions 8, 9, 10, 13, 14, 17, 20, and 21 in the structure.
- Verify total: 23 carbons and 8 chiral atoms match the formula.
Why C is correct:
- Matches the standard formula C23H32O6 with 8 tetrahedral stereocenters as per steroid nomenclature.
Why the others are wrong:
- A: Carbon count is low (22 instead of 23 from missing acetate carbon).
- B: Chiral count is low (7 instead of 8, omitting one side-chain center).
- D: Assumes extra carbon (24 instead of 23, overcounting acetate).
Final answer: C
Topic: An introduction to organic chemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me