A Levels Chemistry (9701)•9701/12/O/N/21
Explanation
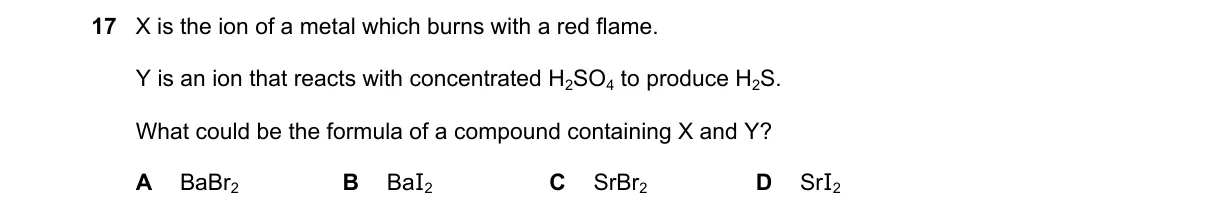
Flame test and H2SO4 reaction identify ions in the compound
Steps:
- X is the metal cation; red flame test identifies Sr^{2+} (strontium), while Ba^{2+} gives green.
- Y is the anion; reaction with concentrated H2SO4 producing H2S occurs with I^-, as HI reduces H2SO4 to H2S, I2, and SO2.
- Br^- with concentrated H2SO4 yields Br2 and SO2, but no H2S.
- Compound containing Sr^{2+} and I^- is SrI2.
Why D is correct:
- Sr^{2+} produces crimson red flame; I^- reacts with concentrated H2SO4 via 8HI + 5H2SO4 → 4I2 + H2S + 4SO2 + 4H2O.
Why the others are wrong:
- A: Ba^{2+} gives apple-green flame, not red.
- B: Ba^{2+} gives apple-green flame, not red.
- C: Br^- produces Br2 and SO2 with concentrated H2SO4, not H2S.
Final answer: D
Topic: Group 2
Practice more A Levels Chemistry (9701) questions on mMCQ.me