A Levels Chemistry (9701)•9701/11/O/N/21
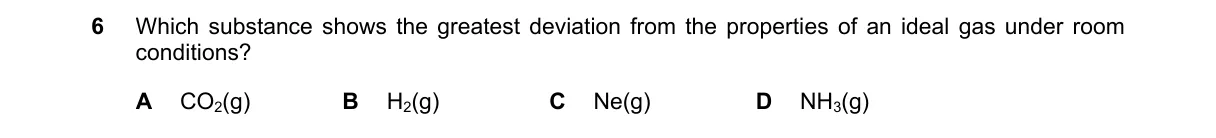
Explanation
Intermolecular forces drive deviations from ideal gas behavior
Steps:
- Ideal gases assume no intermolecular attractions or molecular volume; real gases deviate more with stronger forces or larger sizes at room temperature (25°C, 1 atm).
- Evaluate gases by polarity and bonding: nonpolar/monatomic gases behave ideally; polar gases with hydrogen bonding deviate most.
- Compare options: H₂ and Ne are nonpolar/small; CO₂ has weak dipole-induced forces; NH₃ has strong hydrogen bonding.
- NH₃ shows greatest deviation due to its polarity and intermolecular attractions, confirmed by higher compressibility factor deviation.
Why D is correct:
- NH₃ exhibits strong hydrogen bonding (N-H...N), violating ideal gas law's zero-interaction assumption, leading to largest PV/RT deviation per van der Waals equation.
Why the others are wrong:
- A. CO₂: Linear molecule with weak London dispersion forces, minimal deviation.
- B. H₂: Nonpolar diatomic with negligible forces and small size, nearly ideal.
- C. Ne: Monatomic noble gas with no intermolecular forces, closest to ideal.
Final answer: D
Topic: States of matter
Practice more A Levels Chemistry (9701) questions on mMCQ.me