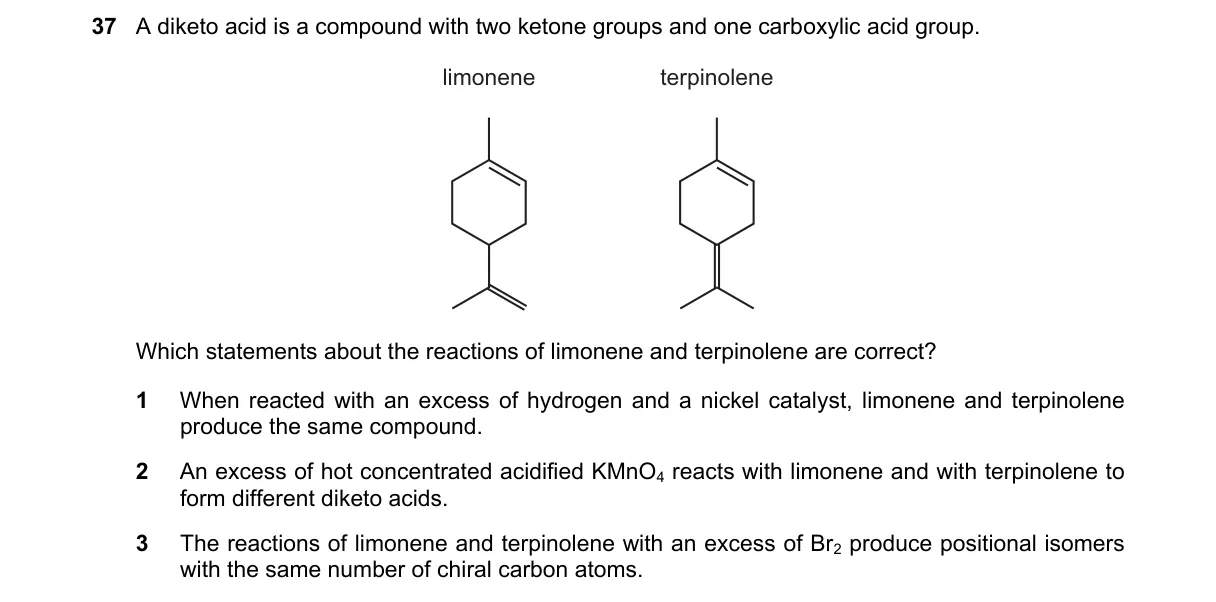
Explanation
Oxidative Cleavage Distinguishes Limonene and Terpinene Steps: - Identify double bond positions: limonene has one endocyclic (ring) and one exocyclic double bond; terpinene has two endocyclic double bonds. - Analyze KMnO4 reaction: hot concentrated acidic KMnO4 cleaves each C=C to carbonyls (ketone for >C= , carboxylic acid for -CH= , CO2 for =CH2). - Predict products for B: limonene yields a diketo acid with ketone from exocyclic and dicarbonyl from ring; terpinene yields a different diketo acid from dual ring cleavages opening to distinct chain. - Verify other statements: hydrogenation saturates both identically; Br2 addition yields non-isomeric tetrabromides with varying chirality. Why B is correct: - KMnO4 cleavage follows Markownikov-like rules for alkenes, producing unique diketo acids due to limonene's exocyclic =CH2 (→ CO2 + ketone) vs. terpinene's symmetric ring diene (→ linear dicarboxylic diketone). Why the others are wrong: - A: Both fully hydrogenate to identical p-menthane (1-isopropyl-4-methylcyclohexane), contradicting the "same compound" claim if interpreted as different. - C: Br2 addition products are constitutional isomers but not positional (different skeletons: exocyclic vs. both endocyclic); chiral carbons differ (limonene: 3; …
Practice more A Levels Chemistry (9701) questions on mMCQ.me