A Levels Chemistry (9701)•9701/11/O/N/21
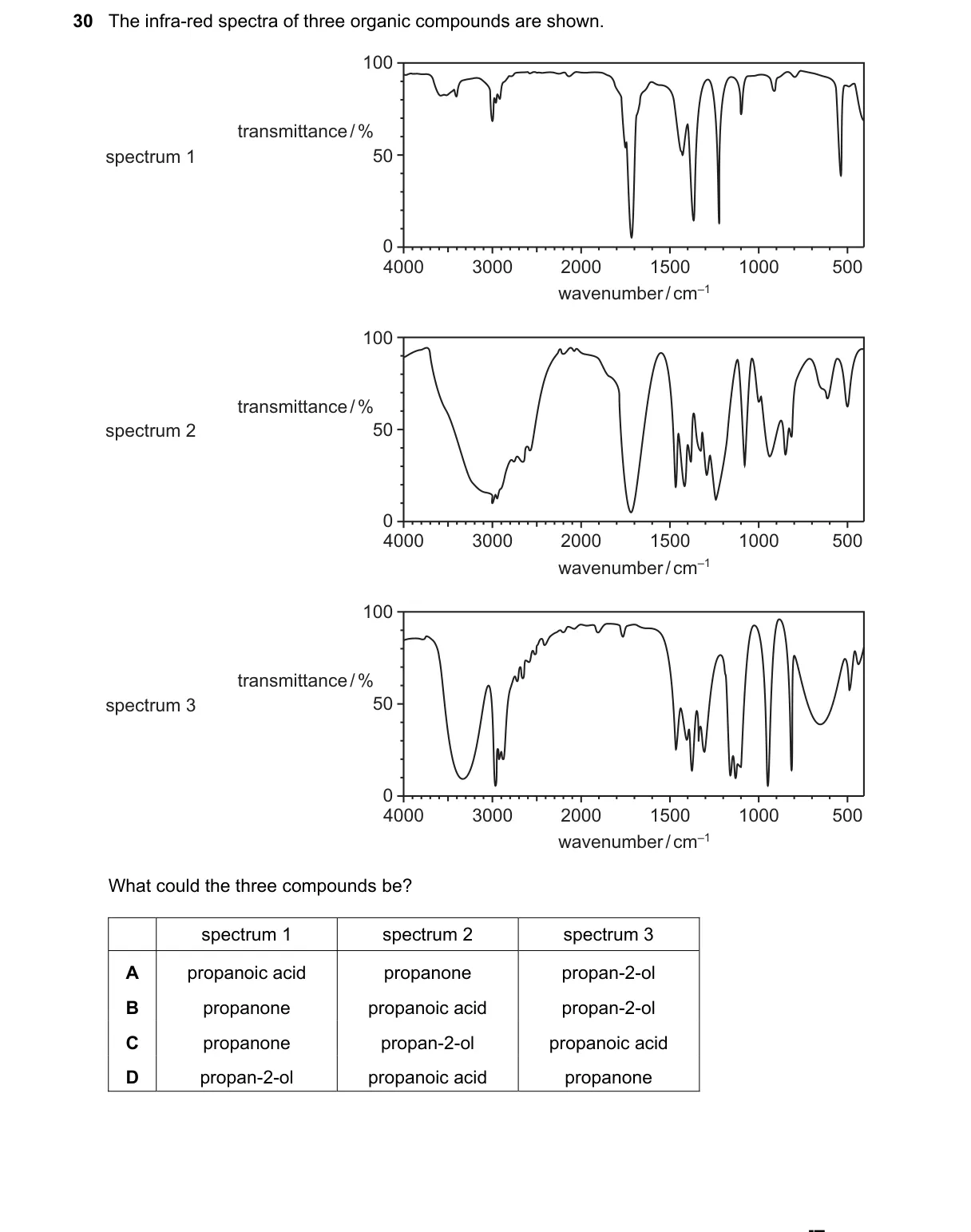
Explanation
IR spectroscopy distinguishes functional groups by characteristic absorption bands
Steps:
- Examine peaks for O-H stretch (broad 3200-3600 cm⁻¹ in alcohols/acids), C=O stretch (1700-1750 cm⁻¹ in carbonyls), and aldehydic C-H (2700-2800 cm⁻¹ doublets in aldehydes).
- Spectrum with C=O at ~1715 cm⁻¹ and no O-H or aldehydic C-H identifies a ketone (propanone).
- Spectrum with C=O at ~1725 cm⁻¹ plus aldehydic C-H doublets identifies an aldehyde (propanal).
- Spectrum with broad O-H at ~3300 cm⁻¹, C-O at ~1100 cm⁻¹, and no C=O identifies a secondary alcohol (propan-2-ol).
Why B is correct:
- Option B matches distinct IR signatures: ketone (C=O only), aldehyde (C=O + aldehydic C-H per IR correlation tables), alcohol (O-H only).
Why the others are wrong:
- A includes carboxylic acid (broad O-H overlapping C=O), indistinguishable from alcohol's O-H.
- C has two alcohols (propan-1-ol and propan-2-ol) with nearly identical broad O-H peaks.
- D includes acid (broad O-H + C=O) and alcohol (O-H), lacking a unique third spectrum.
Final answer: B
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me