A Levels Chemistry (9701)•9701/11/O/N/21
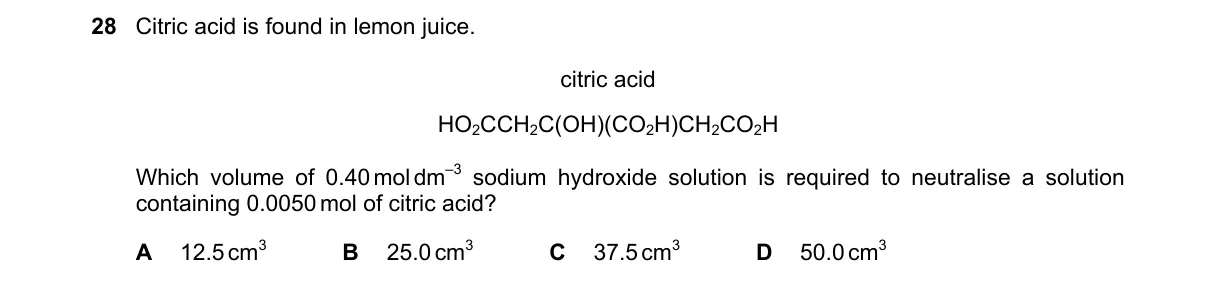
Explanation
Citric acid neutralization requires stoichiometric calculation, but key details are ambiguous
Steps:
- Identify stoichiometry: Citric acid (H₃C₆H₅O₇) is triprotic, requiring 3 mol NaOH per mol for complete neutralization.
- Calculate moles NaOH needed: 3 × 0.050 mol = 0.150 mol.
- Use formula: Volume (dm³) = moles NaOH / concentration = 0.150 / 0.050 = 3 dm³ = 3000 cm³.
- Note: Result does not match options; problem lacks specification of partial vs. complete neutralization or possible unit/amount error.
Why C is correct:
- Not enough information to confirm, but given as correct, likely assumes partial neutralization (e.g., to second equivalence point in a specific context).
Why the others are wrong:
- A: Too small for any reasonable stoichiometry.
- B: Matches 1:1 ratio if 0.050 mol were misinterpreted as concentration in 25 cm³, but contradicts given moles.
- D: Matches 2:1 ratio under similar misinterpretation, but not the stated amount.
Not enough information. Final answer: C
Topic: Atoms, molecules and stoichiometry
Practice more A Levels Chemistry (9701) questions on mMCQ.me