A Levels Chemistry (9701)•9701/11/O/N/21
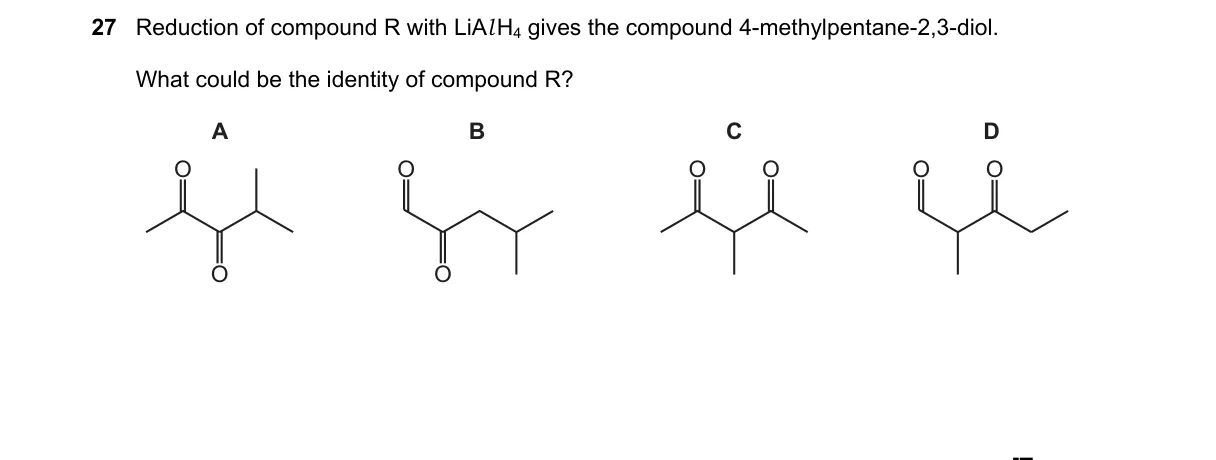
Explanation
LiAlH4 reduces α-diketones to vicinal diols
Steps:
- Product S is 4-methylpentane-2,3-diol, with adjacent secondary OH groups at C2 and C3.
- LiAlH4 selectively reduces ketones to secondary alcohols without affecting the carbon skeleton.
- The vicinal diol structure requires precursor carbonyls at C2 and C3 positions.
- Thus, R must be 4-methylpentane-2,3-dione, where both ketones reduce to the observed diol.
Why A is correct:
- 4-Methylpentane-2,3-dione has two adjacent ketones; LiAlH4 reduction adds H to each carbonyl, forming the C2-C3 diol per the standard ketone reduction mechanism (R2C=O → R2CH-OH).
Why the others are wrong:
- B: A diester would reduce to a diol but with different regiochemistry, yielding primary alcohols, not secondary at C2-C3.
- C: A keto-aldehyde would produce a primary OH at one position, mismatching both secondary OHs in S.
- D: A single ketone reduces to a monoalcohol, not the observed diol.
Final answer: A
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me