A Levels Chemistry (9701)•9701/11/O/N/21
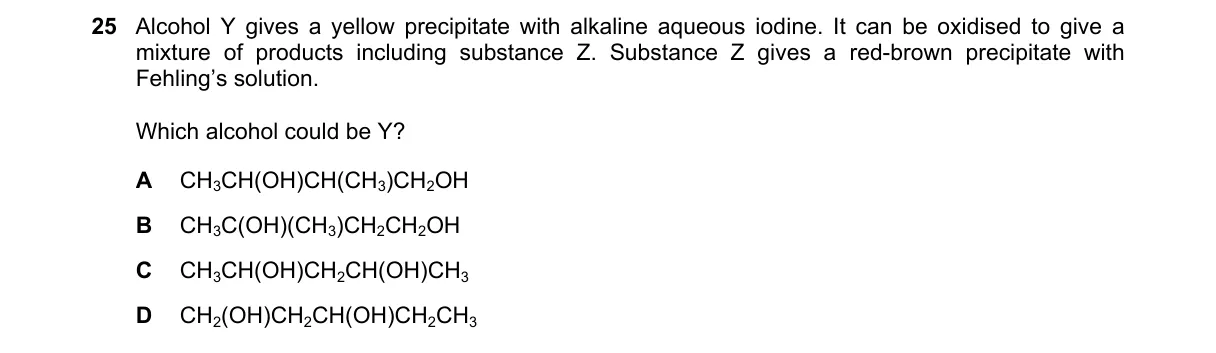
Explanation
Vicinal diol with methyl carbinol group for iodoform and cleavage to aldehyde Steps:
- Yellow precipitate with alkaline I2 shows iodoform test positive for CH3CH(OH)R alcohols.
- Oxidation yields mixture including Z; Z's red-brown Fehling's precipitate indicates aldehyde.
- Eliminate D (ether, no OH for tests); C (butan-2-ol, oxidizes to ketone CH3COCH2CH3, no aldehyde).
- A and B have diol with CH3CH(OH)-; A is vicinal (adjacent OH), cleaves to CH3CHO + HCHO (both aldehydes); B is 1,3-diol (non-adjacent), oxidizes to non-cleaving products like CH3COCH2CH2OH (ketone) + CH3CH(OH)CH2CHO (aldehyde, but no cleavage mixture of simple aldehydes). Why A is correct:
- Propane-1,2-diol's vicinal OH enables oxidative cleavage (e.g., by I2/OH-) to acetaldehyde and formaldehyde, both reducing aldehydes per Fehling's mechanism. Why the others are wrong:
- B: Non-vicinal diol; no cleavage, primary oxidation yields beta-hydroxy aldehyde but lacks defining mixture of simple reducing carbonyls.
- C: Secondary alcohol only; oxidizes to methyl ketone (no Fehling's), single product not mixture.
- D: No alcohol group; fails iodoform and oxidation.
Final answer: A
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me