A Levels Chemistry (9701)•9701/11/O/N/21
Explanation
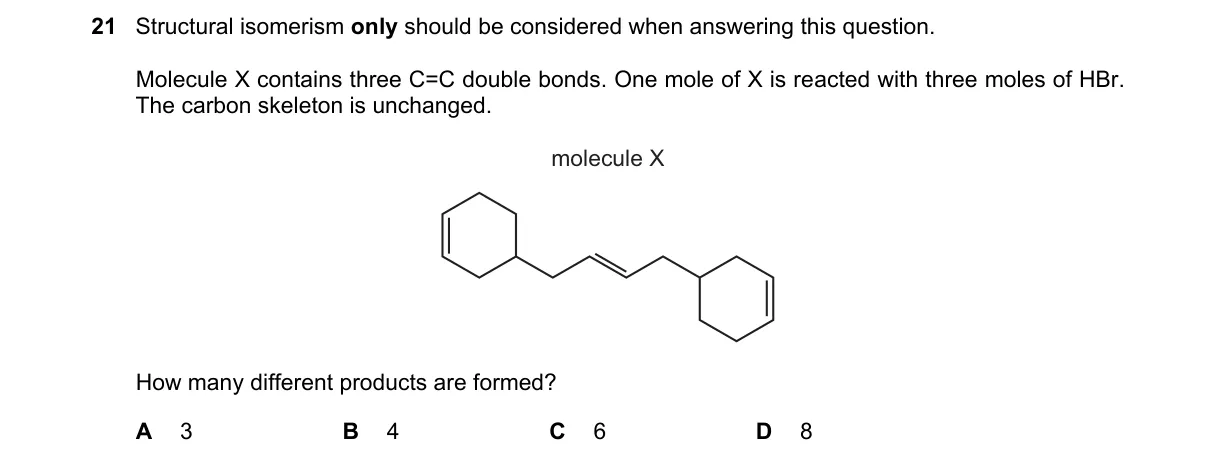
Regioisomers from HBr addition to conjugated triene
Steps:
- Molecule X is 1,3,5-hexatriene (CH₂=CH-CH=CH-CH=CH₂), with three conjugated C=C bonds.
- First HBr adds via protonation at a terminal carbon, forming a delocalized allylic carbocation resonating across the chain (positions 2, 4, 6), yielding three diene intermediates via Br⁻ attack.
- Each diene intermediate undergoes second HBr addition, producing two regioisomers per diene (1,2- and 1,4-addition modes).
- Third HBr addition to the remaining alkene in each case is regioselective (Markovnikov), resulting in four unique structural isomers of the tribromide after accounting for molecular symmetry.
Why B is correct:
- Conjugation enables delocalized carbocations, leading to exactly four distinct positional isomers for the three Br atoms in C₆H₁₁Br₃, per standard polyene addition mechanisms.
Why the others are wrong:
- A. 3: Matches only the first-step intermediates, ignoring further additions.
- C. 6: Counts all second-step products without merging symmetric duplicates.
- D. 8: Assumes two independent regioisomers per isolated double bond (2³), disregarding conjugation.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me