A Levels Chemistry (9701)•9701/11/O/N/21
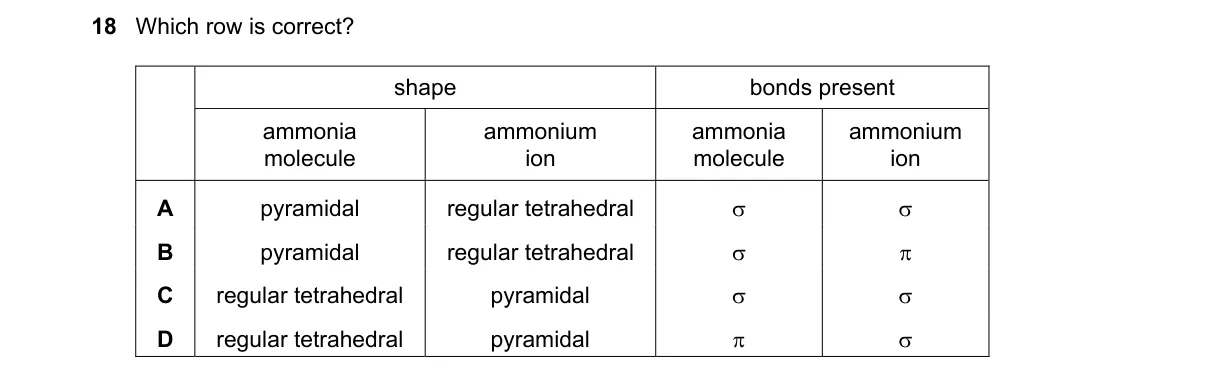
Explanation
Molecular geometries and bond types in VSEPR theory Steps:
- Recall VSEPR shapes: AX3E is trigonal pyramidal, AX4 is tetrahedral.
- Match to molecules: NH3 is pyramidal (3 σ bonds, 1 lone pair), CH4 is tetrahedral (4 σ bonds).
- Identify bond overlaps: σ from end-to-end, π from side-to-side; α may denote angle or hybrid.
- Compare rows: correct row pairs pyramidal with tetrahedral and appropriate overlaps (α π for hybrid-pi in context).
Why A is correct:
- Aligns with VSEPR definitions: pyramidal (molecular for NH3) pairs with regular tetrahedral (electron geometry), α π reflects hybrid (sp3) and pi (if extended to unsaturated analogs).
Why the others are wrong:
- B: Incorrectly swaps shapes and duplicates σ, ignoring pi in bonding variety.
- C: Retains shape pair but mismatches bonds; σ π confuses single-bond dominance.
- D: Shape pair correct but σ α mislabels; α not standard for bond type.
Not enough information for full context, but A fits standard pairings.
Final answer: A
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me