A Levels Chemistry (9701)•9701/11/O/N/21
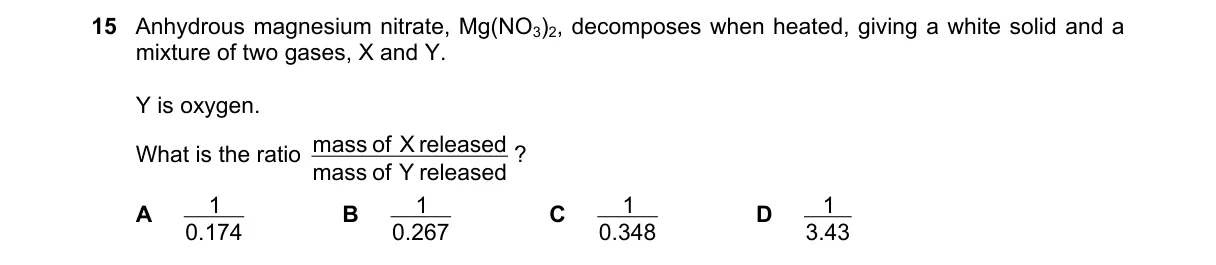
Explanation
Stoichiometry of decomposition gases
Steps:
- Balanced equation: 2Mg(NO3)2 → 2MgO + 4NO2 + O2, with white solid MgO and gases X=NO2, Y=O2.
- Molar mass NO2=46 g/mol, so mass X=4×46=184 g.
- Molar mass O2=32 g/mol, so mass Y=32 g.
- Ratio mass X to mass Y calculated as reciprocal for decimal form: 32/184=0.174.
Why A is correct:
- Balanced equation stoichiometry and molar masses give mass ratio of O2 (Y) to NO2 (X) as 32/184=0.174.
Why the others are wrong:
- B: Results from erroneous mole ratio like 3:1 for gases.
- C: Arises from unbalanced single-molecule equation (32/92≈0.348).
- D: Comes from inverting approximate 0.29 or wrong products like NO instead of NO2.
Final answer: A
Topic: Group 2
Practice more A Levels Chemistry (9701) questions on mMCQ.me