A Levels Chemistry (9701)•9701/13/O/N/20
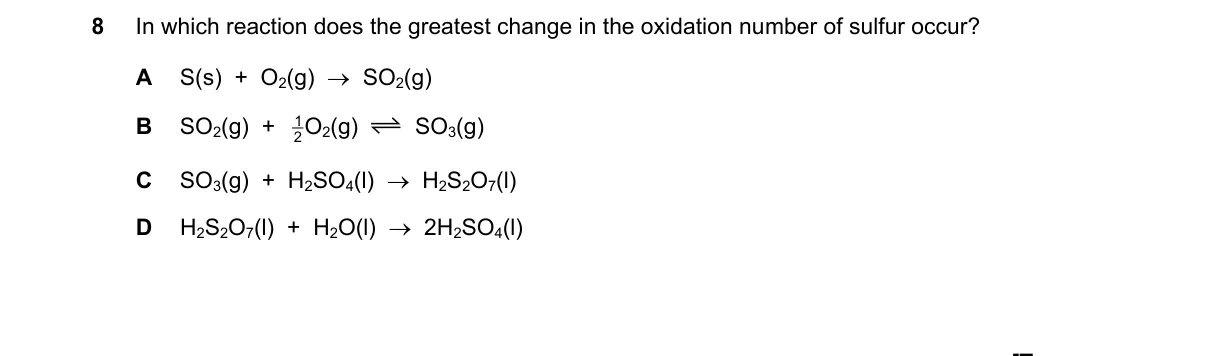
Explanation
Oxidation State Change of Sulfur
Steps:
- Assign oxidation numbers to sulfur in reactants and products for each reaction.
- Calculate the absolute change in oxidation number for sulfur in each case.
- Compare the magnitudes of these changes across options.
- Select the reaction with the largest change.
Why A is correct:
- Sulfur oxidizes from 0 in S(s) to +4 in SO2(g), a change of 4 units, per oxidation number rules where free elements are 0 and O is -2.
Why the others are wrong:
- B: Sulfur changes from +4 in SO2 to +6 in SO3, a smaller change of 2.
- C: Sulfur changes from -2 in H2S to 0 in S(s), a change of 2.
- D: No reaction occurs; H2SO4 remains +6 in both sides, change of 0.
Final answer: A
Topic: Nitrogen and sulfur
Practice more A Levels Chemistry (9701) questions on mMCQ.me