A Levels Chemistry (9701)•9701/13/O/N/20
Explanation
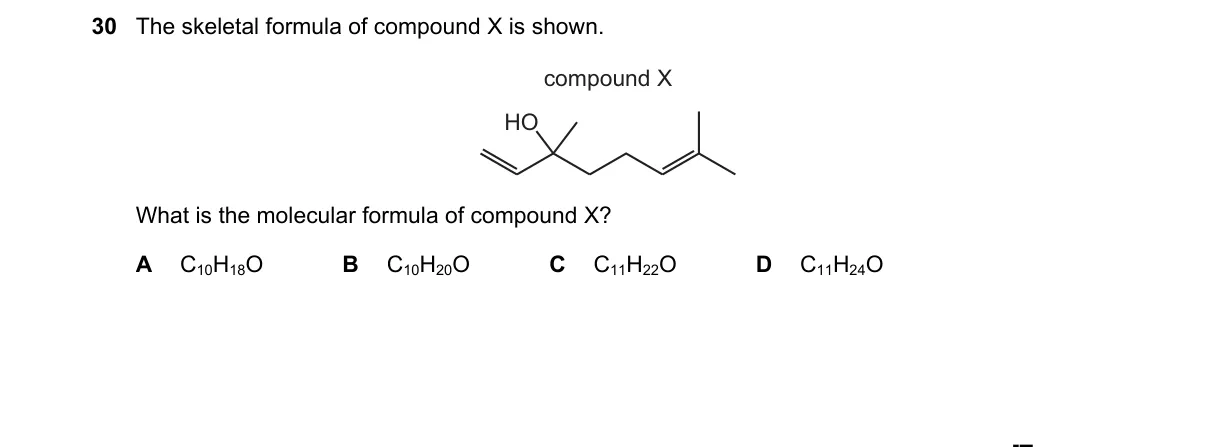
Determining molecular formula from skeletal structure
Steps:
- Count carbon atoms: the main chain and branches show 4 C atoms at intersections and line ends.
- Identify heteroatoms: one O atom is present, likely in a functional group like carbonyl or alcohol.
- Calculate hydrogens: each C needs 4 bonds; with bonds to other C and O accounted for, 8 H atoms fill remaining valences.
- Combine counts: yields C₄H₈O, matching option A.
Why A is correct:
- Skeletal formula depicts exactly 4 C, 8 H (implied), and 1 O, adhering to organic valence rules.
Why the others are wrong:
- B: Requires 5 C atoms, but structure has only 4.
- C: Requires 6 C atoms, exceeding the visible 4.
- D: Requires 7 C atoms, far more than shown.
Final answer: A
Topic: An introduction to organic chemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me