A Levels Chemistry (9701)•9701/13/O/N/20
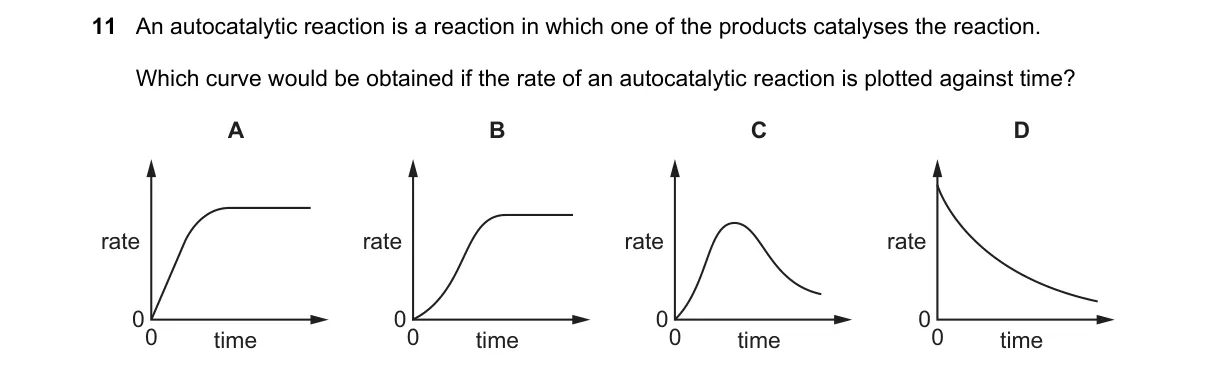
Explanation
Autocatalytic reactions show sigmoidal rate-time profiles
Steps:
- Recall that autocatalysis involves a product accelerating the reaction, starting slow due to low initial catalyst.
- As product accumulates, rate increases rapidly, forming an inflection point.
- Eventually, reactant depletion causes the rate to level off or decline.
- Plot rate vs. time yields a sigmoidal (S-shaped) curve matching option C.
Why C is correct:
- The sigmoidal shape reflects the rate law for autocatalysis, where rate = k [reactant][product], leading to exponential product growth initially then saturation.
Why the others are wrong:
- A: Linear curve implies constant rate, typical of zero-order kinetics, not autocatalysis.
- B: Decreasing curve suggests inhibition or depletion without acceleration, opposite of autocatalysis.
- D: Exponential rise without plateau ignores reactant limits, unrealistic for finite systems.
Final answer: C
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me