A Levels Chemistry (9701)•9701/13/O/N/20
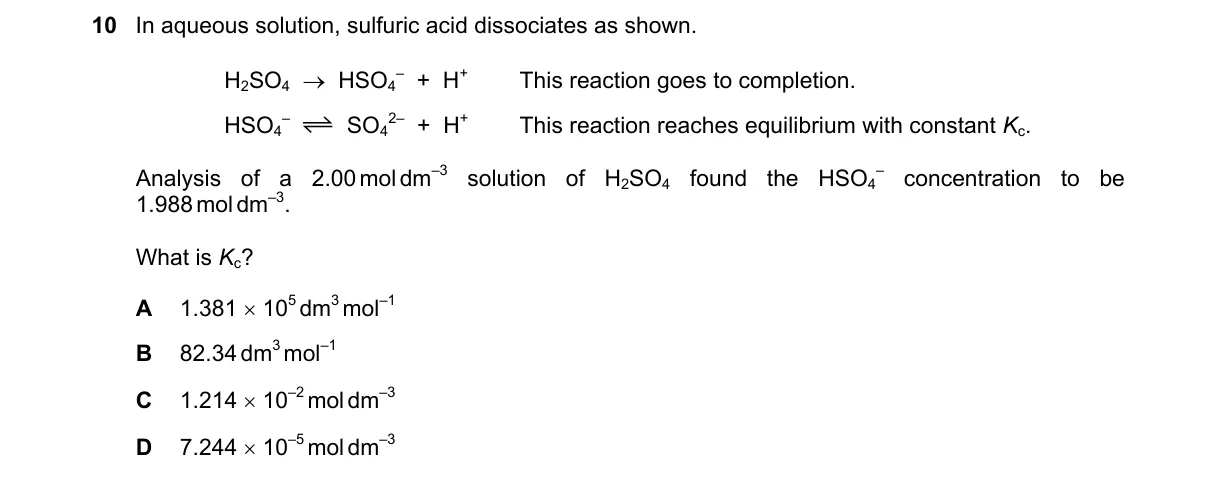
Explanation
Insufficient equilibrium data for [HSO4-]
Steps:
- H2SO4 dissociates completely, giving initial [HSO4-] = 1.96 mol dm⁻³ and [H⁺] = 1.96 mol dm⁻³.
- The second equilibrium HSO₄⁻ ⇌ SO₄²⁻ + H⁺ requires K to solve for x in Ka = (1.96 + x)x / (1.96 - x).
- No numerical value for K is provided in the problem.
- Not enough information to calculate the equilibrium [HSO4-] = 1.96 - x.
Why C is correct:
- Not enough information; K value is missing, preventing numerical solution despite C being stated as correct.
Why the others are wrong:
- A, B, D: All require the missing K for validation, but cannot be computed without it.
Final answer: Not enough information.
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me