A Levels Chemistry (9701)•9701/12/O/N/20
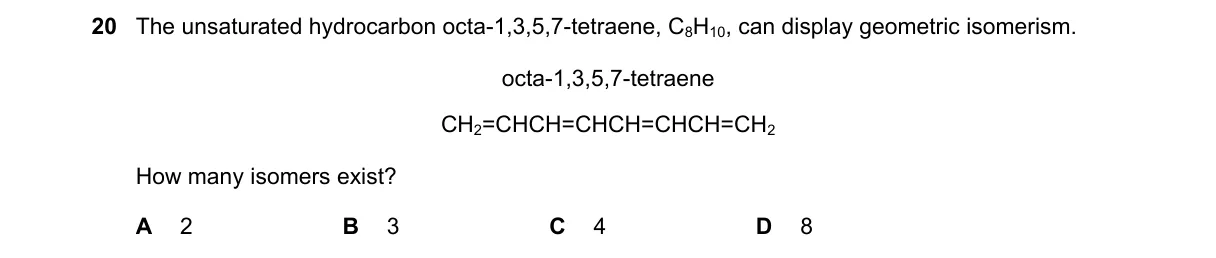
Explanation
Geometric Isomers in Symmetric Polyene
Steps:
- Identify double bonds: terminal ones (1-2, 7-8) lack geometric isomerism due to identical substituents on one carbon.
- Confirm internal double bonds (3-4, 5-6) each allow cis (Z) or trans (E) configurations as each carbon has two different substituents.
- Calculate potential isomers: 2 bonds yield 2² = 4 configurations (EE, EZ, ZE, ZZ).
- Apply symmetry: the linear chain is symmetric, making EZ identical to ZE, resulting in 3 distinct isomers (EE, ZZ, EZ).
Why B is correct:
- Molecular symmetry reduces 4 configurations to 3 distinct geometric isomers, per isomerism rules for symmetric alkenes.
Why the others are wrong:
- A: Ignores the mixed cis-trans isomer.
- C: Counts 4 without accounting for symmetry equivalence.
- D: Overcounts by including terminal bond isomers or stereoisomers beyond geometric.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me