A Levels Chemistry (9701)•9701/11/O/N/20
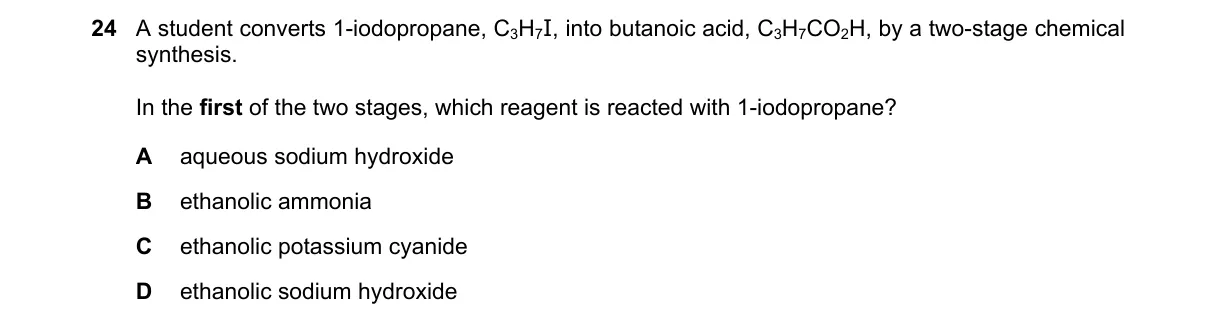
Explanation
Nucleophilic substitution with cyanide to extend carbon chain
Steps:
- Butanoic acid (C3H7CO2H) requires adding one carbon to the propyl chain from 1-iodopropane (C3H7I).
- Primary alkyl iodides undergo SN2 substitution with nucleophiles to replace I.
- CN- from ethanolic KCN acts as nucleophile, forming propanenitrile (C3H7CN).
- Hydrolysis of the nitrile in the second stage yields butanoic acid.
Why C is correct:
- Ethanolic KCN delivers CN- for SN2 reaction, converting C3H7I to C3H7CN per the general formula R-X + KCN → R-CN + KI.
Why the others are wrong:
- A: Aqueous NaOH substitutes to form propan-1-ol (C3H7OH), not extending the chain.
- B: Ethanolic ammonia substitutes to form propan-1-amine (C3H7NH2), yielding an amine.
- D: Ethanolic NaOH favors elimination to propene or substitution to propan-1-ol, not chain extension.
Final answer: C
Topic: Organic synthesis
Practice more A Levels Chemistry (9701) questions on mMCQ.me