A Levels Chemistry (9701)•9701/13/O/N/19
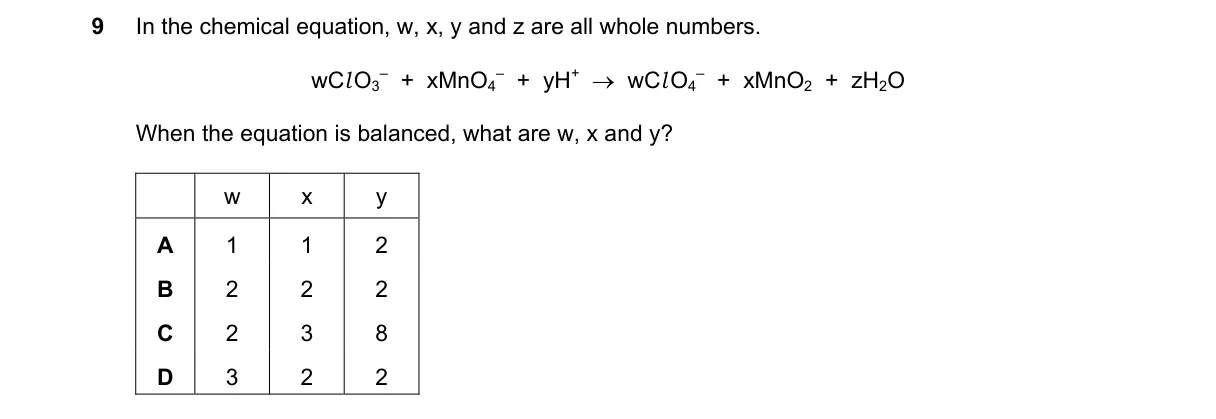
Explanation
Balancing redox reaction via half-reaction method in acidic medium Steps:
- Oxidation: ClO₃⁻ + H₂O → ClO₄⁻ + 2H⁺ + 2e⁻ (Cl from +5 to +7)
- Reduction: MnO₄⁻ + 4H⁺ + 3e⁻ → MnO₂ + 2H₂O (Mn from +7 to +4)
- Equalize electrons by multiplying oxidation by 3 and reduction by 2 (6e⁻ each)
- Combine and simplify: 3ClO₃⁻ + 2MnO₄⁻ + 2H⁺ → 3ClO₄⁻ + 2MnO₂ + H₂O (cancels 3H₂O and 6H⁺) Why D is correct:
- Coefficients 3, 2, 2 match 3×2e⁻ lost = 2×3e⁻ gained, conserving electrons per the redox law. Why the others are wrong:
- A: 1×2e⁻ ≠ 1×3e⁻, electrons imbalanced
- B: 2×2e⁻ ≠ 3×3e⁻, electrons imbalanced
- C: Identical to B, electrons imbalanced
Final answer: D
Topic: Chemistry of transition elements
Practice more A Levels Chemistry (9701) questions on mMCQ.me