A Levels Chemistry (9701)•9701/13/O/N/19
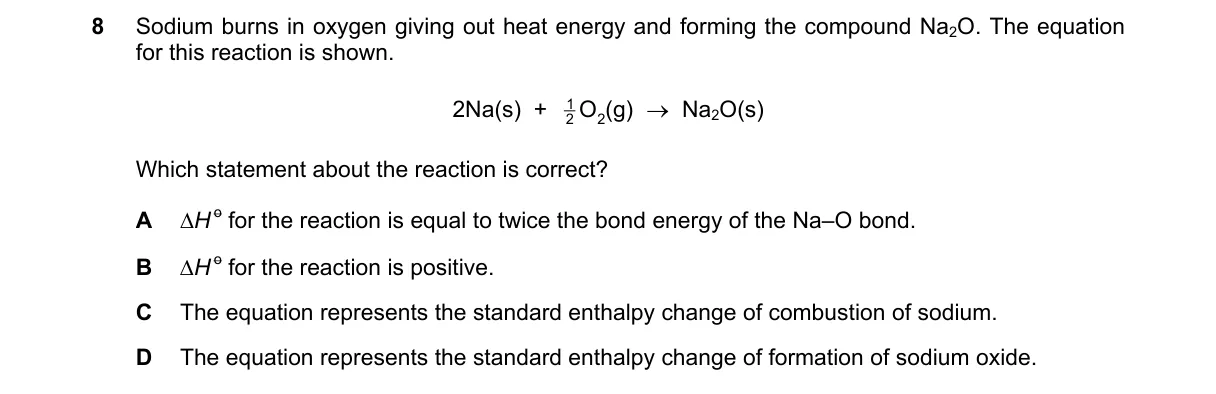
Explanation
Reaction Represents Formation of Na₂O from Elements
Steps:
- Reactants are Na(s) and O₂(g), the standard states of sodium and oxygen.
- Product is 1 mole of Na₂O(s).
- Standard enthalpy of formation (ΔH_f°) is the ΔH when 1 mole of a compound forms from elements in standard states.
- This equation matches the definition of ΔH_f° for Na₂O.
Why D is correct:
- ΔH_f° is defined as the enthalpy change for forming 1 mole of a compound from its elements in standard states at 298 K and 1 atm, exactly as shown.
Why the others are wrong:
- A: ΔH accounts for breaking O=O bond and forming ionic lattice in Na₂O, not just one Na-O bond energy.
- B: ΔH involves multiple energy terms (O=O breaking, lattice formation), not simply twice a single bond energy.
- C: ΔH_c° for sodium is per mole of Na (Na + ½O₂ → ½Na₂O), not for 1 mole of Na₂O formation.
Final answer: D
Topic: Chemical energetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me