A Levels Chemistry (9701)•9701/13/O/N/19
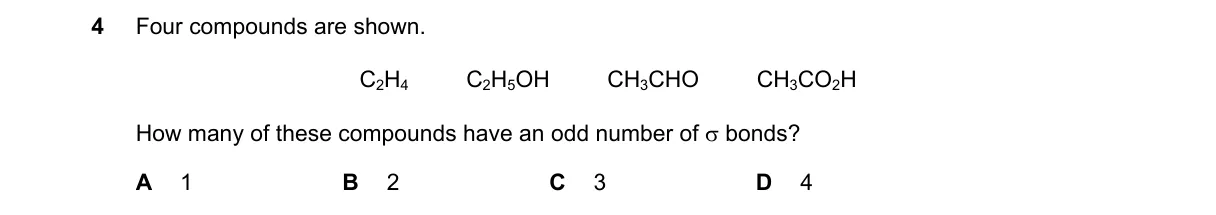
Explanation
Counting total covalent bonds in each compound
Steps:
- CH3 (methyl radical): 3 C-H single bonds, total 3 (odd).
- CH3OH (methanol): 3 C-H, 1 C-O, 1 O-H single bonds, total 5 (odd).
- CH3CHO (acetaldehyde): 3 C-H (methyl), 1 C-C, 1 C-H (aldehyde), 1 C=O sigma + 1 C=O pi, total 8 (even).
- CH2OH (hydroxymethyl radical): 2 C-H, 1 C-O, 1 O-H single bonds, total 4 (even).
Why B is correct:
- Exactly two compounds (CH3 and CH3OH) have an odd total number of covalent bonds, per Lewis structure bond counting where multiple bonds contribute fully.
Why the others are wrong:
- A is wrong: Two compounds have odd bonds, not one.
- C is wrong: Only two have odd bonds, not three.
- D is wrong: Two have even bonds, so not all four.
Final answer: B
Topic: An introduction to organic chemistry
Practice more A Levels Chemistry (9701) questions on mMCQ.me