A Levels Chemistry (9701)•9701/13/O/N/19
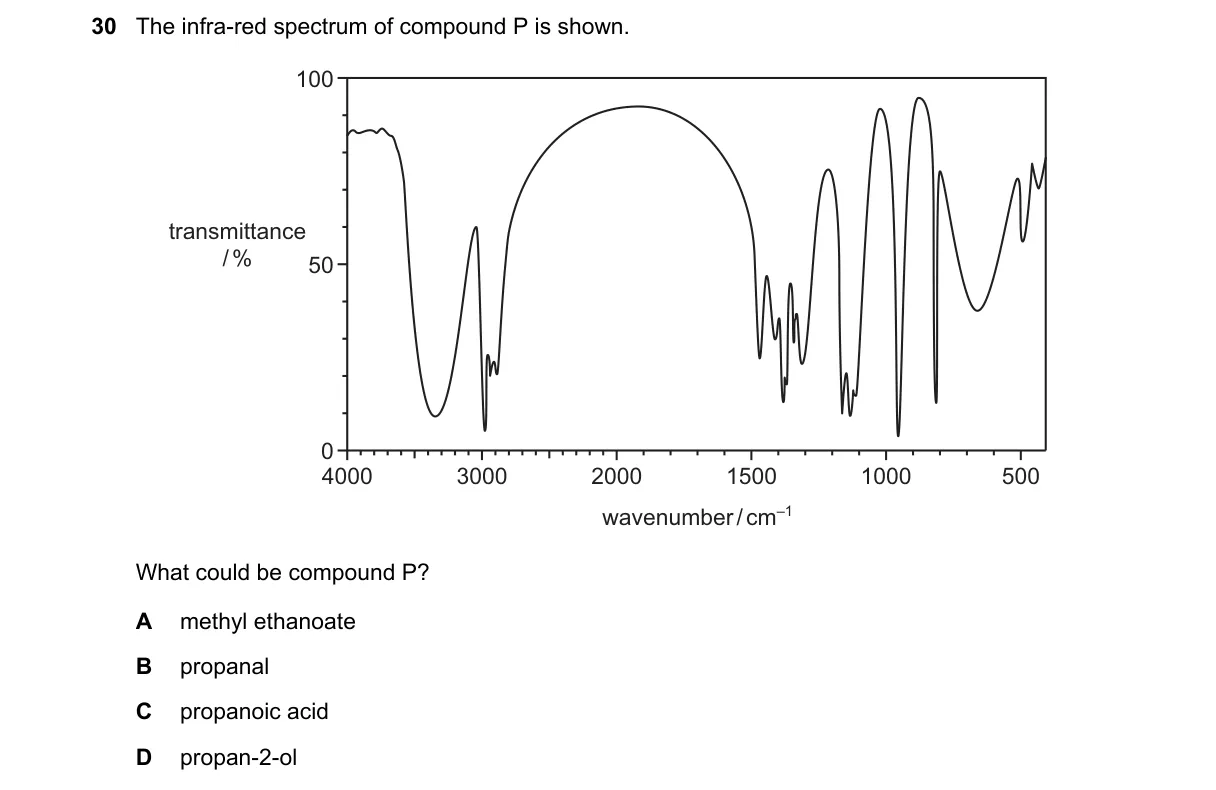
Explanation
IR spectrum identifies functional groups via characteristic absorption bands
Steps:
- Examine the spectrum for a broad absorption around 3200-3600 cm⁻¹, indicating O-H stretch from hydrogen bonding.
- Check for absence of a sharp carbonyl (C=O) peak near 1700-1750 cm⁻¹, ruling out carbonyl-containing compounds.
- Look for C-O stretch around 1000-1200 cm⁻¹, supporting alcohol presence.
- Confirm no aldehyde C-H stretches (2700-2800 cm⁻¹) or acidic O-H broadening (2500-3300 cm⁻¹).
Why D is correct:
- Propan-2-ol (CH₃CH(OH)CH₃) shows a broad O-H stretch at 3200-3600 cm⁻¹ due to hydrogen bonding in alcohols and a C-O stretch at 1000-1200 cm⁻¹, with no C=O band.
Why the others are wrong:
- A: Methyl ethanoate has a strong C=O ester stretch at 1735 cm⁻¹.
- B: Propanal exhibits C=O aldehyde stretch at 1720 cm⁻¹ and C-H aldehyde bands at 2700-2800 cm⁻¹.
- C: Propanoic acid displays broad O-H acid stretch at 2500-3300 cm⁻¹ and C=O at 1710 cm⁻¹.
Final answer: D
Topic: Analytical techniques
Practice more A Levels Chemistry (9701) questions on mMCQ.me