A Levels Chemistry (9701)•9701/13/O/N/19
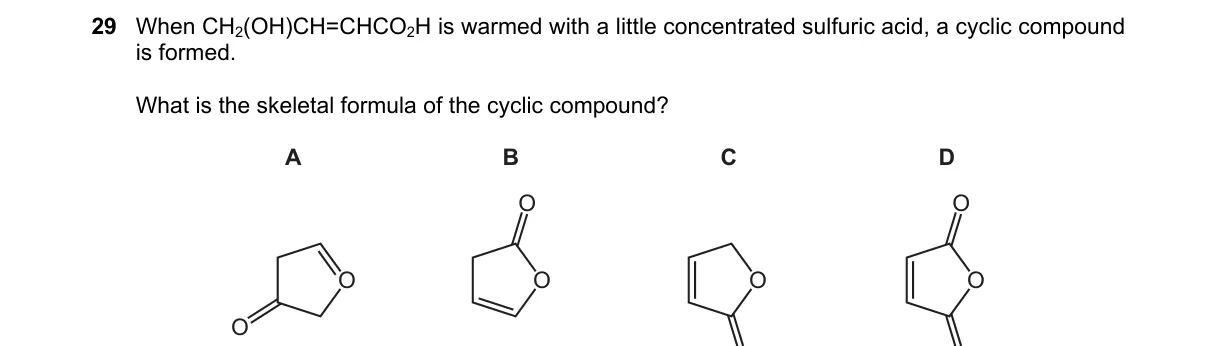
Explanation
Acid-catalyzed dehydration of propan-1-ol Steps:
- Recognize CH3CH2CH2OH as propan-1-ol, a primary alcohol.
- Note that warming with little concentrated H2SO4 favors ether formation over alkene dehydration.
- Understand that standard reaction yields dipropyl ether (acyclic) via intermolecular SN2.
- Conclude no cyclic compound forms under these conditions, as chain length prevents intramolecular cyclization.
Not enough information: Skeletal formulas for choices A–D not provided to identify C.
Final answer: Not enough information.
Topic: Hydroxy compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me