A Levels Chemistry (9701)•9701/13/O/N/19
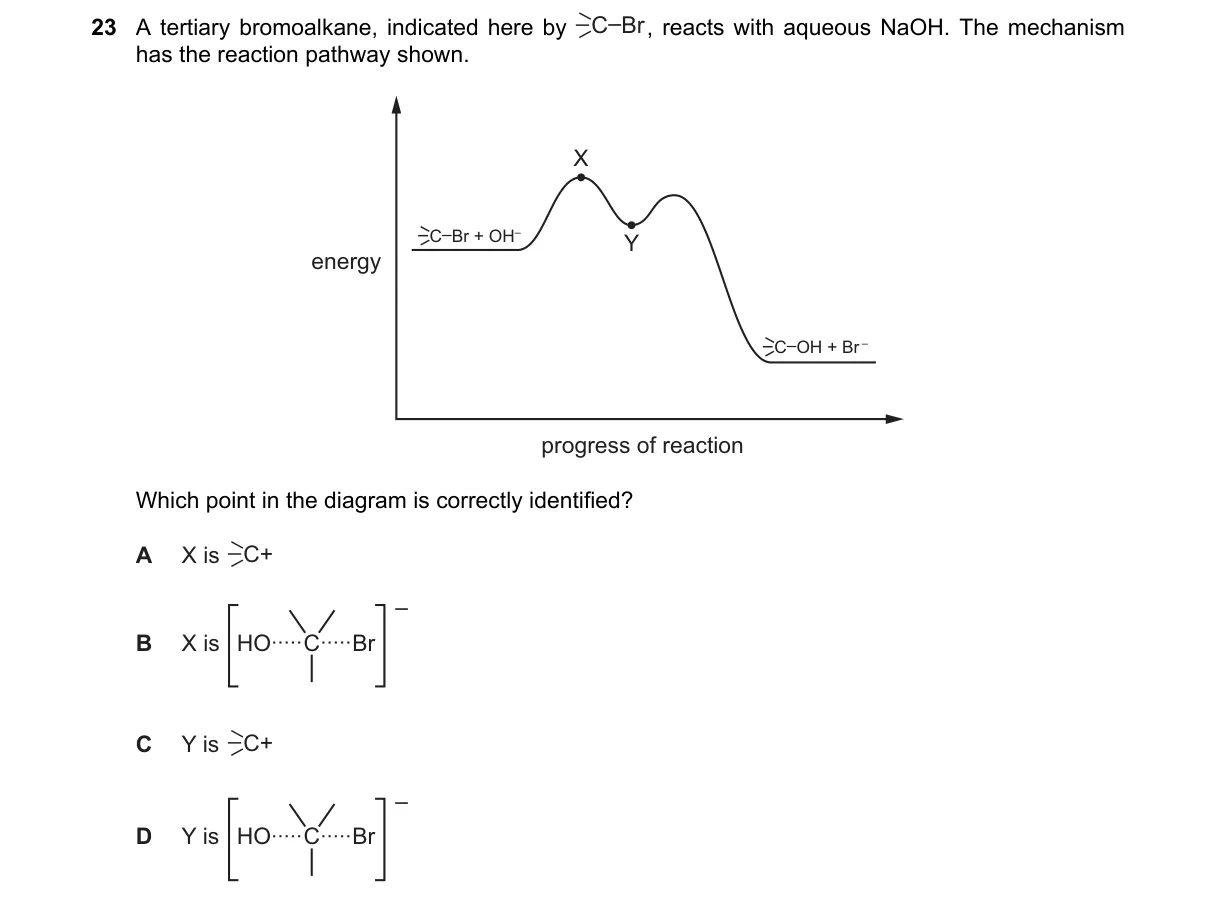
Explanation
SN1 Mechanism Intermediate Identification
Steps:
- Tertiary bromoalkane favors SN1 with aqueous NaOH due to stable tertiary carbocation formation.
- Rate-determining step: heterolytic cleavage of C-Br bond, producing R₃C⁺ intermediate and Br⁻.
- Energy diagram shows initial transition state (X), then carbocation minimum (Y).
- Nucleophile OH⁻ then attacks Y to form R₃C-OH.
Why C is correct:
- Y corresponds to the carbocation (C⁺) intermediate, the energy minimum after Br⁻ departure in SN1 pathway.
Why the others are wrong:
- A: X is the transition state for ionization, not the stable C⁺.
- B: HO-C-Br represents no known SN1 species; it's not formed.
- D: Y is the carbocation, not the protonated alcohol-like HO-C-Br.
Final answer: C
Topic: Halogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me