A Levels Chemistry (9701)•9701/13/O/N/19
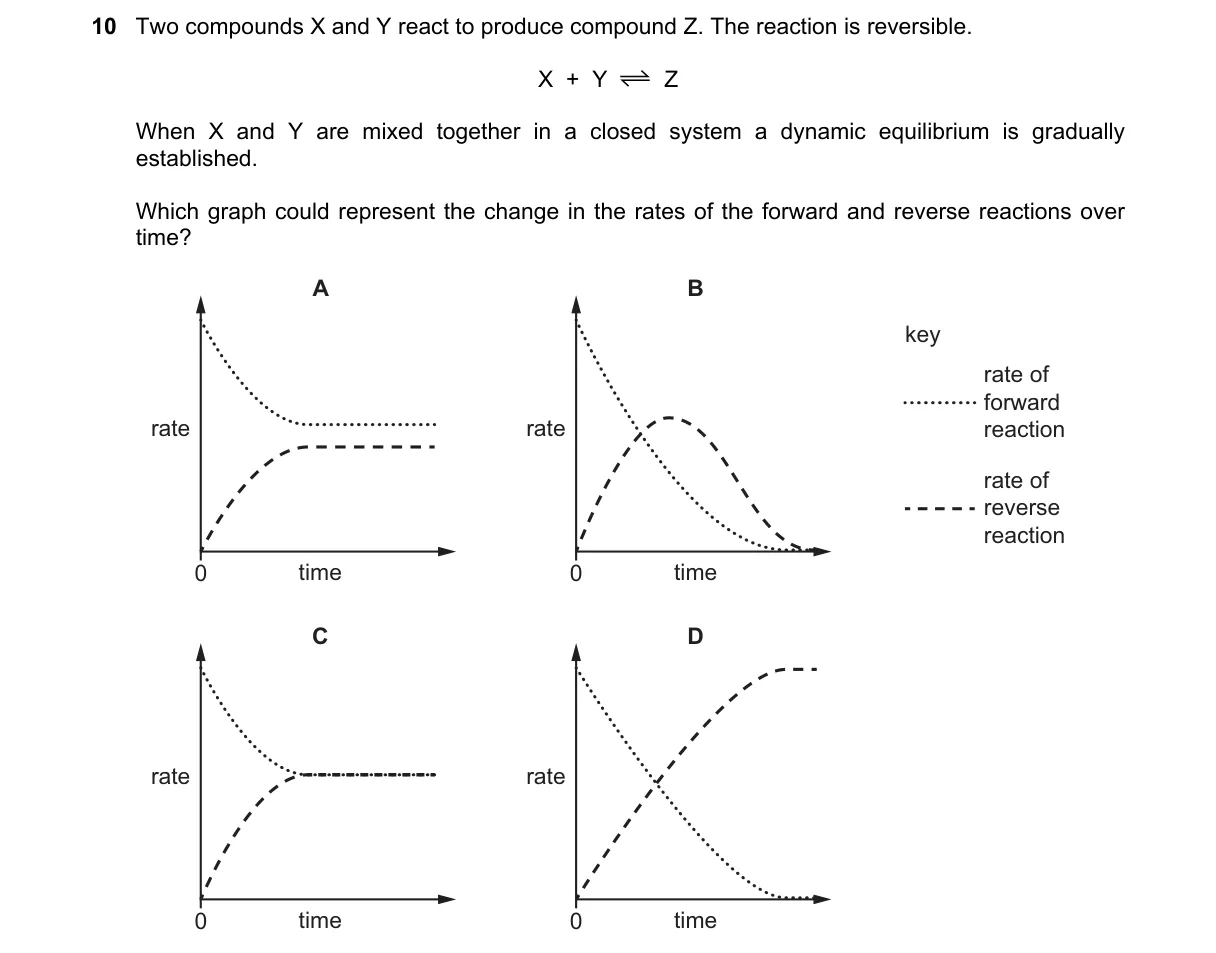
Explanation
Dynamic equilibrium occurs when forward and reverse reaction rates become equal and constant.
Steps:
- At t=0, only forward reaction proceeds, so its rate is maximum while reverse rate is zero.
- As Z accumulates, reverse rate increases proportionally to [Z].
- Forward rate decreases as [X] and [Y] diminish.
- Rates intersect and remain equal thereafter, establishing equilibrium.
Why C is correct:
- Graph C depicts forward rate decreasing and reverse rate increasing until they meet and level off, matching the definition of dynamic equilibrium where net reaction stops but reactions continue at equal rates.
Why the others are wrong:
- A: Both rates constant from start, ignoring initial absence of products.
- B: Forward rate unchanged while reverse rises, violating reactant depletion.
- D: Rates cross but continue changing, not stabilizing at equality.
Final answer: C
Topic: Equilibria
Practice more A Levels Chemistry (9701) questions on mMCQ.me