A Levels Chemistry (9701)•9701/13/O/N/19
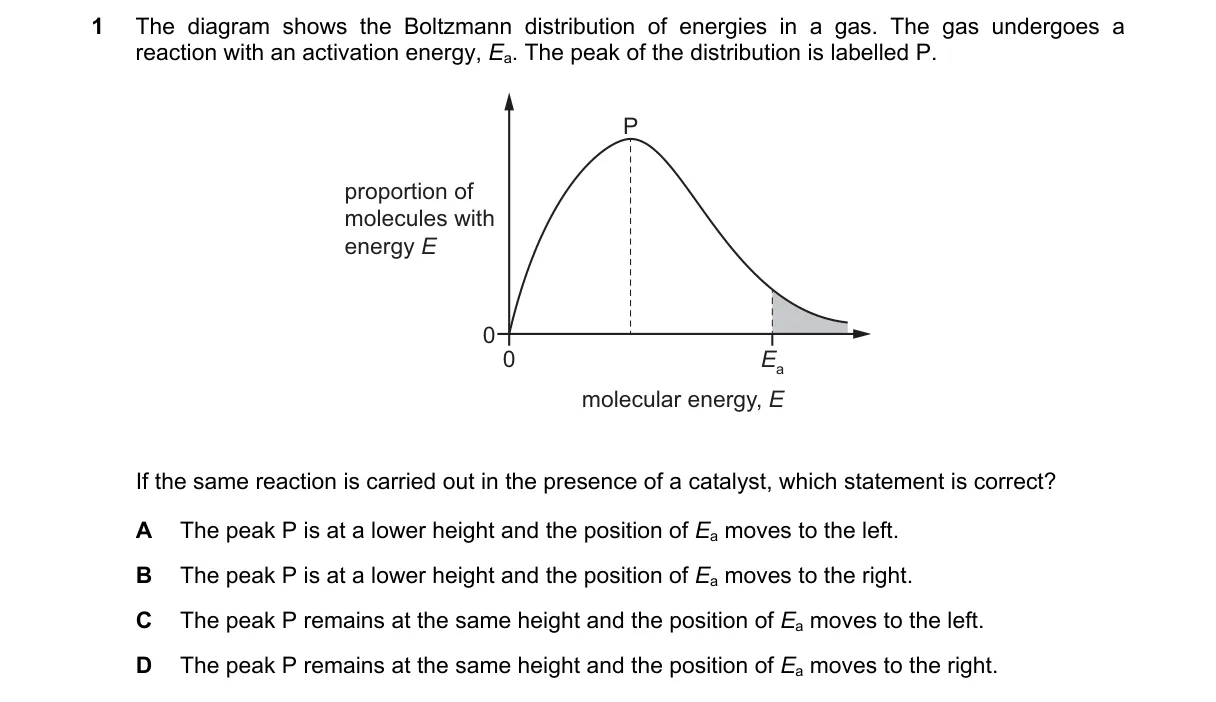
Explanation
Catalysts lower activation energy without altering molecular energy distribution Steps:
- Catalysts provide a pathway with reduced activation energy E_a, increasing reaction rate.
- Maxwell-Boltzmann distribution shows energy distribution of molecules, fixed by temperature.
- Peak of distribution (most probable energy) stays same height and position with catalyst.
- E_a line shifts left to lower energy on x-axis (energy increasing rightward), increasing molecules above E_a.
Why B is correct:
- Collision theory states catalysts decrease E_a (moving its position left), leaving distribution curve unchanged per Boltzmann distribution f(E) = (2/√π)(E^{1/2}/(kT)^{3/2}) exp(-E/kT).
Why the others are wrong:
- A: Peak height unchanged; catalyst does not modify energy distribution.
- C: E_a moves left (lower), not right (higher E_a would slow reaction).
- D: Peak height and E_a position both unaffected or wrongly shifted.
Final answer: B
Topic: Reaction kinetics
Practice more A Levels Chemistry (9701) questions on mMCQ.me