A Levels Chemistry (9701)•9701/12/O/N/19
Explanation
Empirical formula simplifies molecular formula ratios Steps:
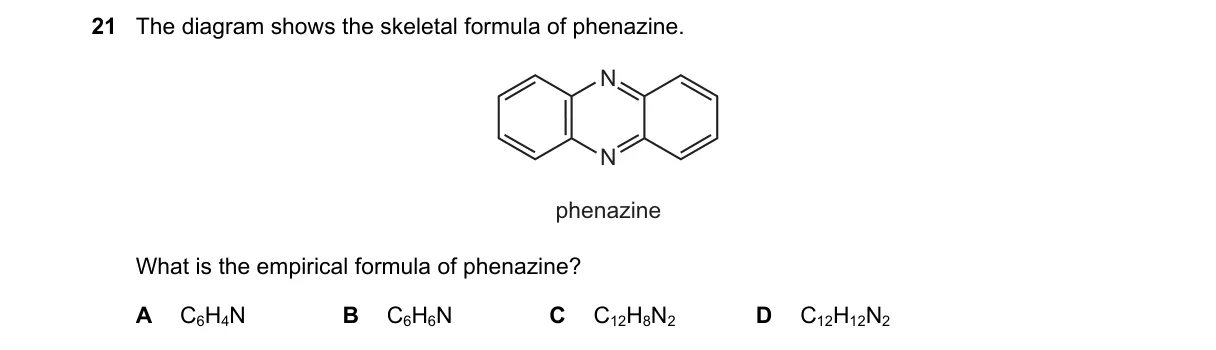
- Examine the skeletal formula of phenazine, a tricyclic aromatic compound with two fused benzene rings and a central pyrazine ring.
- Count atoms: 12 carbon, 8 hydrogen, 2 nitrogen atoms.
- Obtain molecular formula C₁₂H₈N₂.
- Divide subscripts by greatest common divisor (2) to get simplest ratio: C₆H₄N.
Why B is correct:
- Empirical formula is the lowest whole-number ratio of elements; C₁₂H₈N₂ reduces to C₆H₄N by dividing by 2.
Why the others are wrong:
- A: C₄H₄N ratio (4:4:1) does not match phenazine's 6:4:1 atom ratio.
- C: C₁₂H₈N₂ is the full molecular formula, not simplified.
- D: C₁₂H₈N omits one nitrogen, undercounting heteroatoms in the structure.
Final answer: B
Topic: Nitrogen compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me