A Levels Chemistry (9701)•9701/13/O/N/18
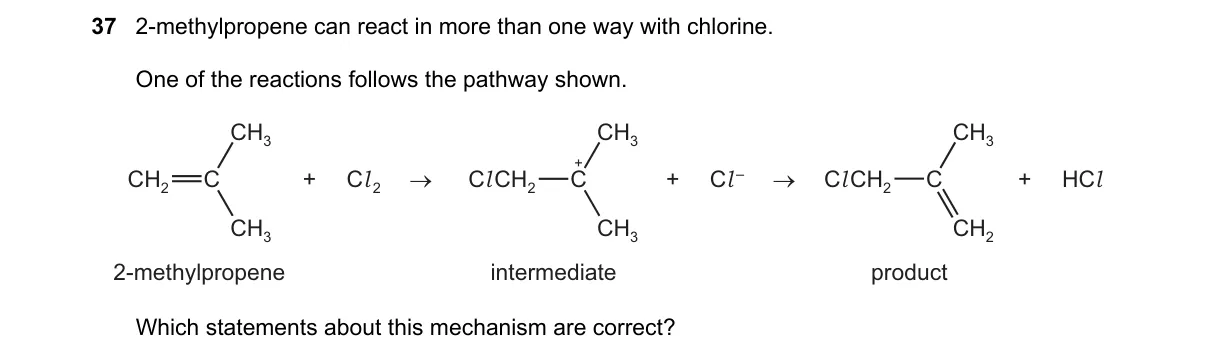
Explanation
Electrophilic Addition Mechanism for Alkene Halogenation
Steps:
- The π electrons of 2-methylpropene attack Cl₂, forming a chloronium ion intermediate and releasing Cl⁻.
- The chloronium ion bridges the double bond, with positive charge more on the tertiary carbon due to stability.
- Cl⁻ attacks the more substituted tertiary carbon from the opposite side, leading to anti addition.
- The product is 2-chloro-2-methyl-1-chloropropane, following Markovnikov's rule for unsymmetrical alkenes.
Why B is correct:
- B accurately describes the chloronium ion as a three-membered ring intermediate, per the standard mechanism for halogen addition to alkenes.
Why the others are wrong:
- A incorrectly suggests a free radical pathway, which requires light or initiators not mentioned.
- C wrongly implies a carbocation intermediate without bridging, leading to syn addition possibilities.
- D misstates the regioselectivity, as Cl⁻ attacks the tertiary carbon, not the primary.
Final answer: B
Topic: Hydrocarbons
Practice more A Levels Chemistry (9701) questions on mMCQ.me