A Levels Chemistry (9701)•9701/13/O/N/18
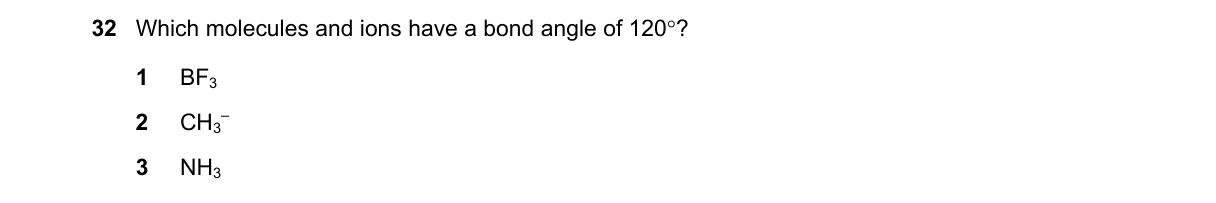
Explanation
VSEPR Theory Predicts Bond Angles from Electron Geometry
Steps:
- Apply VSEPR to count electron domains: bonding and lone pairs determine geometry.
- BF3 has 3 bonding pairs, 0 lone pairs on B (AX3): trigonal planar, 120° angles.
- CH3+ has 3 bonding pairs, 0 lone pairs on C (AX3): trigonal planar, 120° angles.
- NH4+ has 4 bonding pairs, 0 lone pairs on N (AX4): tetrahedral, 109.5° angles.
- Identify option listing both BF3 and CH3+ as having 120° angles.
Why D is correct:
- D selects BF3 and CH3+, both AX3 types with trigonal planar geometry and 120° bond angles per VSEPR.
Why the others are wrong:
- A: Lists only BF3; incomplete, as CH3+ also has 120°.
- B: Lists only CH3+; incomplete, as BF3 also has 120°.
- C: NH4+ is AX4, tetrahedral with 109.5° angles, not 120°.
Final answer: D
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me