A Levels Chemistry (9701)•9701/13/O/N/18
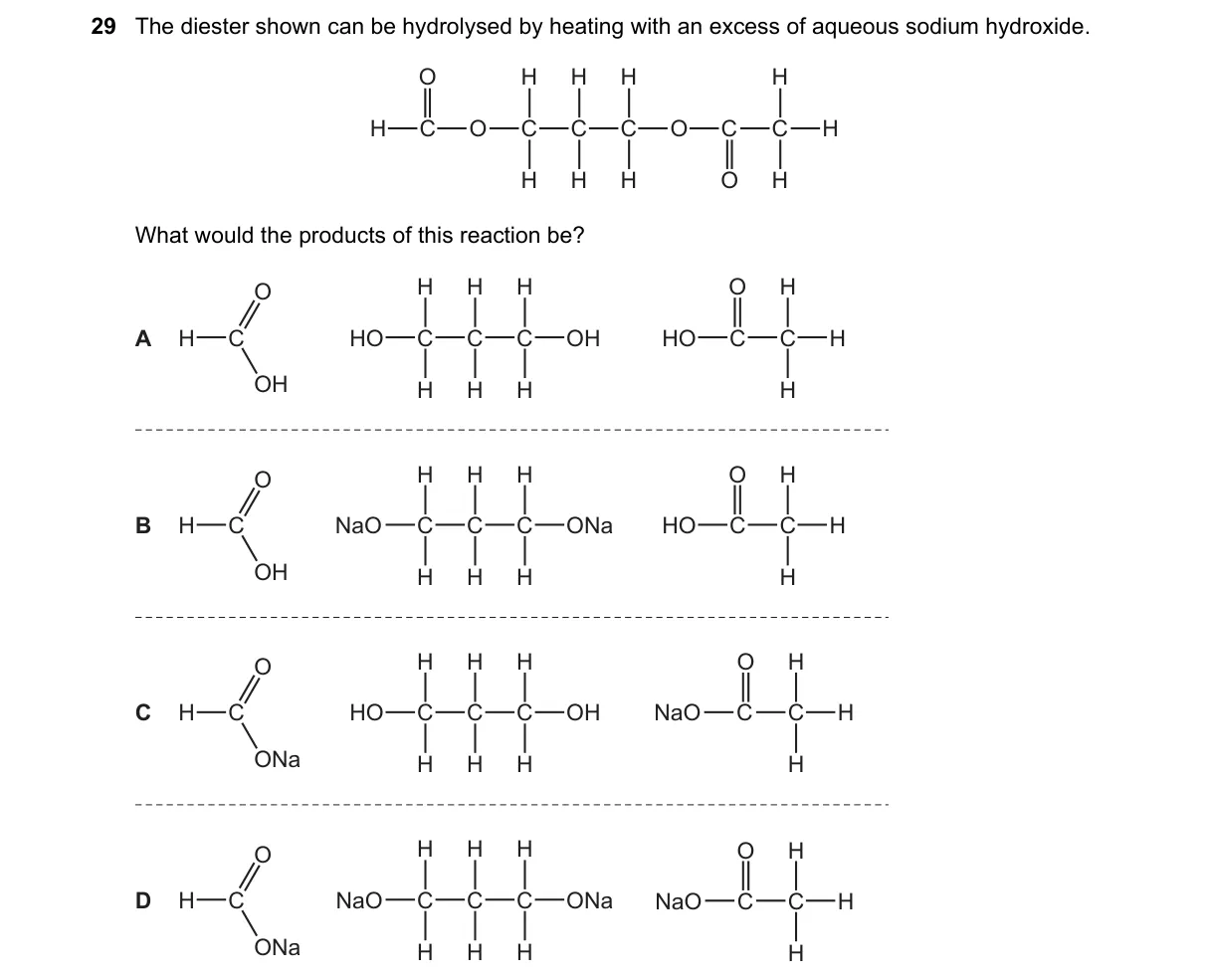
Explanation
Saponification of diester with excess NaOH
Steps:
- Recognize the compound as a diester with two -COOR groups.
- Alkaline hydrolysis cleaves each ester bond using OH⁻ from NaOH.
- Each ester yields a carboxylate salt (-COONa) and an alcohol (ROH).
- Excess NaOH ensures complete conversion to salts, not acids.
Why C is correct:
- C depicts the disodium dicarboxylate and two alcohol molecules, matching the saponification formula: RCOOR' + NaOH → RCOONa + R'OH for both esters.
Why the others are wrong:
- A shows the free diacid, but excess base forms salts, not H⁺-protonated acids.
- B indicates mono-hydrolysis product, ignoring excess NaOH for full reaction.
- D includes unchanged ester or wrong byproducts, violating complete hydrolysis.
Final answer: C
Topic: Carboxylic acids and derivatives
Practice more A Levels Chemistry (9701) questions on mMCQ.me