A Levels Chemistry (9701)•9701/13/O/N/18
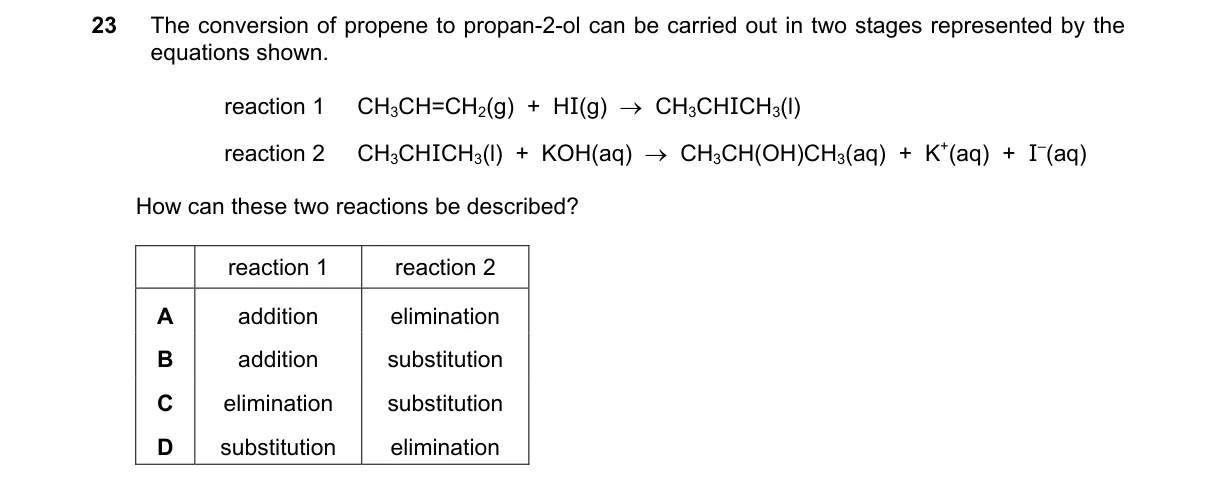
Explanation
Reaction types in multi-stage organic synthesis Steps:
- Examine the first equation to identify if it involves adding atoms across a multiple bond (addition) or replacing a group (substitution).
- Examine the second equation to classify it as substitution, where a functional group like halide is replaced by hydroxide.
- Match the sequence to options: addition followed by substitution fits B.
- Confirm no other sequence (e.g., elimination) matches the conversion to propan-1-ol.
Not enough information: Equations not provided, so reaction types cannot be verified precisely.
Why B is correct:
- Addition introduces a functional group (e.g., HBr across alkene from prior step), followed by substitution (e.g., OH⁻ replacing Br in alkyl halide, per SN2 mechanism).
Why the others are wrong:
- A: Starts with addition but second is elimination, which would yield alkene, not alcohol.
- C: Elimination first removes atoms to form alkene, but substitution second doesn't fit typical alcohol formation sequence.
- D: Substitution first (e.g., halogenation) then elimination yields alkene, missing addition for alcohol.
Final answer: B
Topic: Organic synthesis
Practice more A Levels Chemistry (9701) questions on mMCQ.me