A Levels Chemistry (9701)•9701/13/O/N/18
Explanation
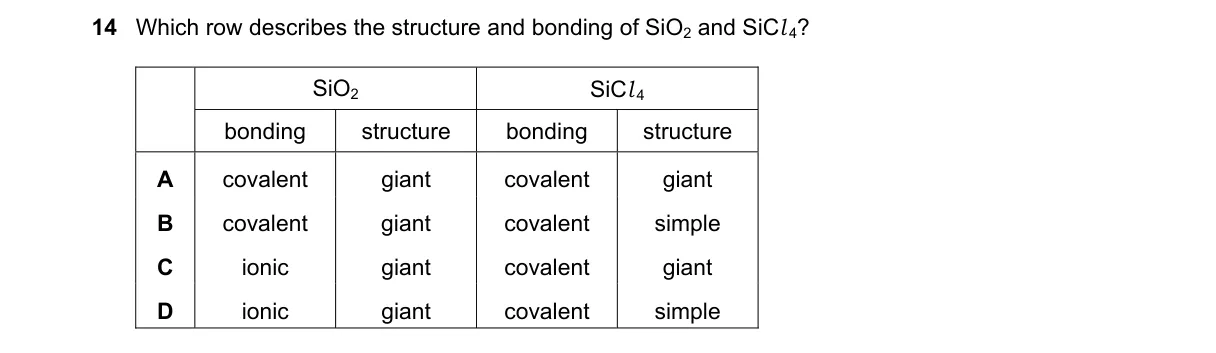
Structure and bonding in silicon oxides and chlorides
Steps:
- Identify SiO₂ as a network solid with silicon-oxygen covalent bonds forming a giant lattice, similar to diamond.
- Determine SiCl₄ as a discrete tetrahedral molecule with polar covalent Si-Cl bonds, existing as simple molecules.
- Confirm both compounds involve covalent bonding, not ionic, due to similar electronegativities between Si and O/Cl.
- Match to option with covalent giant for SiO₂ and covalent simple for SiCl₄.
Why B is correct:
- SiO₂ forms a giant covalent structure via tetrahedral SiO₄ units sharing oxygen atoms, while SiCl₄ is a simple covalent molecule with four Si-Cl bonds, per VSEPR theory.
Why the others are wrong:
- A: SiCl₄ is simple molecular, not giant.
- C: Neither compound is ionic; silicon does not form stable ions like metals.
- D: SiO₂ bonding is covalent, not ionic.
Final answer: B
Topic: Chemical bonding
Practice more A Levels Chemistry (9701) questions on mMCQ.me