A Levels Chemistry (9701)•9701/14/M/J/25
Explanation
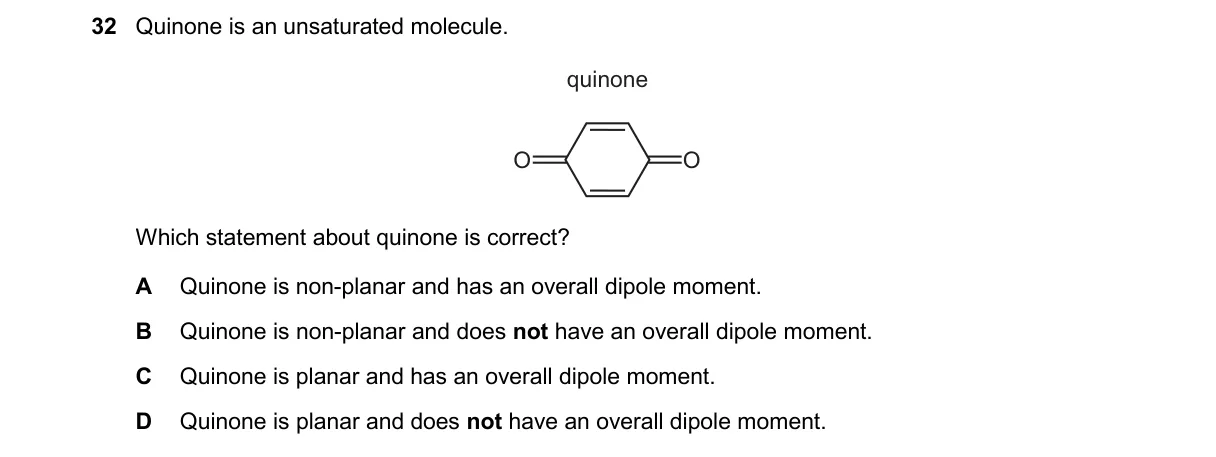
Quinone's Planar Structure and Molecular Symmetry
Steps:
- Quinone (p-benzoquinone) features a six-membered carbon ring with two opposite C=O groups and double bonds.
- All atoms are sp2 hybridized, enforcing a flat, planar geometry for conjugation.
- The molecule has a center of symmetry bisecting the C=O bonds and a plane of symmetry through the ring.
- Bond dipoles from C=O groups cancel due to symmetry, yielding zero net dipole moment.
Why D is correct:
- Quinone's symmetric, conjugated structure ensures planarity (via sp2 hybridization) and no overall dipole (dipole moments cancel per symmetry principles).
Why the others are wrong:
- A: Quinone is planar, not non-planar.
- B: Quinone lacks an overall dipole moment due to symmetry.
- C: Quinone is planar, not non-planar.
Final answer: D
Topic: Carbonyl compounds
Practice more A Levels Chemistry (9701) questions on mMCQ.me